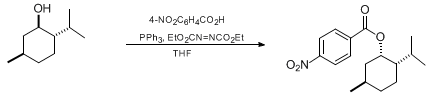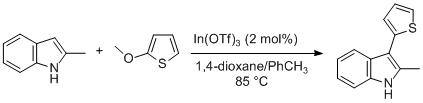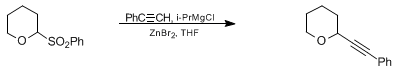Browse · Reactions
Reactions
14 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| A GENERAL PROCEDURE FOR MITSUNOBU INVERSION OF STERICALLY HINDERED ALCOHOLS: INVERSION OF MENTHOL. (1S,2S,5R)-5-METHYL-2-(1-METHYLETHYL)CYCLOHEXYL 4-NITROBENZOATE (Cyclohexanol, 5-methyl-2-(1-methylethyl)-, 4-nitrobenzoate, [1S-(1α,2α,5β)]-) Substitution | Preparation of Cyclohexanol, 5-methyl-2-(1-methylethyl)-, 4-nitrobenzoate, [1S-(1α,2α,5β)]-. Submitted by Jeffrey A. Dodge, Jeffrey S. Nissen, and Misti Presnell1. DOI: 10.15227/orgsyn.073.0110. | Organic Syntheses | |
| Appel reaction Substitution | The Appel reaction is an organic reaction that converts an alcohol into an alkyl chloride using triphenylphosphine and carbon tetrachloride. The use of carbon tetrabromide or bromine as a halide sourc… | Wikipedia: Category:Name reactions | |
| Catalytic Intramolecular Friedel-Crafts Reaction of Benzyl Meldrum's Acid Derivatives: Preparation of 5,6-Dimethoxy-2-Methyl-1-Indanone Electrophilic aromatic substitution | Preparation of Catalytic Intramolecular Friedel-Crafts Reaction of Benzyl Meldrum's Acid Derivatives: Preparation of 5,6-Dimethoxy-2-Methyl-1-Indanone. Submitted by Tiantong Lou, E-Ting Liao, Ashraf W… | Organic Syntheses | |
| Finkelstein reaction Substitution | The Finkelstein reaction, named after the German chemist Hans Finkelstein, is a type of SN2 reaction (substitution nucleophilic bimolecular reaction) that involves the exchange of one halogen atom for… | Wikipedia: Category:Name reactions | |
| Friedel-Crafts acylation: benzene + acetyl chloride → acetophenone Scale Electrophilic aromatic substitution | Textbook electrophilic aromatic substitution via the AlCl3-generated acylium ion. The product ketone (acetophenone) deactivates the ring toward further acylation, so unlike F-C alkylation, monoacylati… | Friedel-Crafts 1877 | |
| Indium-Catalyzed Heteroaryl–Heteroaryl Bond Formation through Nucleophilic Aromatic Substitution: Preparation of 2-Methyl-3-(thien-2-yl)-1H-indole Substitution | Preparation of Indium-Catalyzed Heteroaryl–Heteroaryl Bond Formation through Nucleophilic Aromatic Substitution: Preparation of 2-Methyl-3-(thien-2-yl)-1H-indole. Submitted by Yuta Nagase and Teruhisa… | Organic Syntheses | |
| Mitsunobu reaction Substitution | The Mitsunobu reaction is an organic reaction that converts an alcohol into a variety of functional groups, such as an ester, using triphenylphosphine and an azodicarboxylate such as diethyl azodicarb… | Wikipedia: Category:Name reactions | |
| Mitsunobu reaction: benzyl alcohol + phthalimide → N-benzyl phthalimide Scale Substitution | PPh3/DIAD-mediated dehydrative coupling of an alcohol with an acidic pronucleophile (pKa ≲ 13). Phthalimide as the nucleophile gives an N-alkyl phthalimide that hydrolyses to the free primary amine — … | Mitsunobu 1967 | |
| NUCLEOPHILIC AROMATIC SUBSTITUTION OF ARYL FLUORIDES BY SECONDARY NITRILES: PREPARATION OF 2-(2-METHOXYPHENYL)-2-METHYLPROPIONITRILE (Benzeneacetonitrile, 2-methoxy-α,α-dimethyl-) Substitution | Preparation of Benzeneacetonitrile, 2-methoxy-α,α-dimethyl-. Submitted by Stéphane Caron1 , Jill M. Wojcik, and Enrique Vazquez. DOI: 10.15227/orgsyn.079.0209. | Organic Syntheses | |
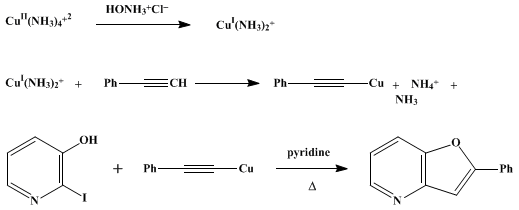
| SUBSTITUTION OF ARYL HALIDES WITH COPPER(I) ACETYLIDES: 2-PHENYLFURO[3,2-b]PYRIDINE (Furo[3,2-b]pyridine, 2-phenyl-) Substitution | Preparation of Furo[3,2-b]pyridine, 2-phenyl-. Submitted by D. C. Owsley and C. E. Castro1. DOI: 10.15227/orgsyn.052.0128. | Organic Syntheses | |
| SUBSTITUTION REACTIONS OF 2-BENZENESULFONYL CYCLIC ETHERS: TETRAHYDRO-2-(PHENYLETHYNYL)-2H-PYRAN (2H-Pyran, tetrahydro-2-(phenylethenyl)-) Substitution | Preparation of 2H-Pyran, tetrahydro-2-(phenylethenyl)-. Submitted by Dearg S. Brown and Steven V. Ley1. DOI: 10.15227/orgsyn.070.0157. | Organic Syntheses | |
| Vicarious nucleophilic substitution Substitution | In organic chemistry, the vicarious nucleophilic substitution is a special type of nucleophilic aromatic substitution in which a nucleophile replaces a hydrogen atom on the aromatic ring and not leavi… | Wikipedia: Category:Name reactions | |
| Williamson ether synthesis Substitution | The Williamson ether synthesis is an organic reaction, forming an ether from an organohalide and a deprotonated alcohol (alkoxide). This reaction was developed by Alexander Williamson in 1850. Typica… | Wikipedia: Category:Name reactions | |
| Williamson ether synthesis: sodium ethoxide + 1-bromobutane → ethyl butyl ether Scale Substitution | Textbook SN2 ether-formation. Sodium ethoxide (typically generated in situ from NaH + EtOH or used as a solid) attacks the primary alkyl bromide via backside attack; the bromide leaves as NaBr, precip… | Williamson 1850 |