Browse · Reactions
Reactions
32 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| 3-HYDROXY-3-METHYL-1-PHENYL-1-BUTANONE BY CROSSED ALDOL REACTION (1-Butanone, 3-hydroxy-3-methyl-1-phenyl-) Condensation | Preparation of 1-Butanone, 3-hydroxy-3-methyl-1-phenyl-. Submitted by Teruaki Mukaiyama and Koichi Narasaka1. DOI: 10.15227/orgsyn.065.0006. | Organic Syntheses | |
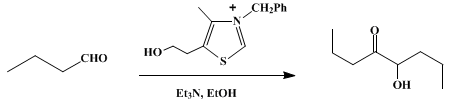
| ACYLOIN CONDENSATION BY THIAZOLIUM ION CATALYSIS: BUTYROIN (4-Octanone, 5-hydroxy-) Condensation | Preparation of 4-Octanone, 5-hydroxy-. Submitted by H. Stetter and H. Kuhlmann1. DOI: 10.15227/orgsyn.062.0170. | Organic Syntheses | |
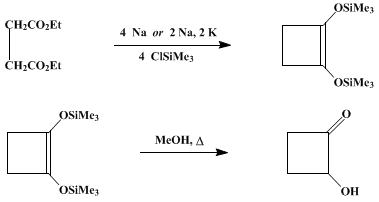
| ACYLOIN CONDENSATION IN WHICH CHLOROTRIMETHYLSILANE IS USED AS A TRAPPING AGENT: 1,2-BIS(TRIMETHYLSILYLOXY)CYCLOBUTENE AND 2-HYDROXYCYCLOBUTANONE (Trimethylsilane, 1-cyclobuten-1,2-ylenedioxybis- and Cyclobutanone, 2-hydroxy-) Condensation | Preparation of Trimethylsilane, 1-cyclobuten-1,2-ylenedioxybis- and Cyclobutanone, 2-hydroxy-. Submitted by Jordan J. Bloomfield1 and Janice M. Nelke. DOI: 10.15227/orgsyn.057.0001. | Organic Syntheses | |
| Aldol–Tishchenko reaction Condensation | The Aldol–Tishchenko reaction is a tandem reaction involving an aldol reaction and a Tishchenko reaction. In organic synthesis, it is a method to convert aldehydes and ketones into 1,3-hydroxyl compou… | Wikipedia: Category:Name reactions | |
| AMIDE FORMATION BY DECARBOXYLATIVE CONDENSATION OF HYDROXYLAMINES AND α-KETOACIDS: N-[(1S)-1 PHENYLETHYL]-BENZENEACETAMIDE Condensation | Preparation of AMIDE FORMATION BY DECARBOXYLATIVE CONDENSATION OF HYDROXYLAMINES AND α-KETOACIDS: N-[(1S)-1 PHENYLETHYL]-BENZENEACETAMIDE. Submitted by Lei Ju and Jeffrey W. Bode1. DOI: 10.15227/orgsy… | Organic Syntheses | |
| ANTI-SELECTIVE BORON-MEDIATED ASYMMETRIC ALDOL REACTION OF CARBOXYLIC ESTERS: SYNTHESIS OF (2S, 3R)-2,4-DIMETHYL-1,3-PENTANEDIOL (1,3-Pentanediol, 2,4-dimethyl-, [S-(R*,S*)-) Condensation | Preparation of 1,3-Pentanediol, 2,4-dimethyl-, [S-(R*,S*)-. Submitted by Atsushi Abiko1. DOI: 10.15227/orgsyn.079.0116. | Organic Syntheses | |
| CATALYTIC ASYMMETRIC ACYL HALIDE-ALDEHYDE CYCLOCONDENSATION REACTION ((4S)-4-(2-Phenylethyl)-2-oxetanone) Condensation | Preparation of (4S)-4-(2-Phenylethyl)-2-oxetanone. Submitted by Scott G. Nelson and Paul M. Mills1. DOI: 10.15227/orgsyn.082.0170. | Organic Syntheses | |
| CATALYTIC ASYMMETRIC SYNTHESIS OF NITROALDOLS USING A LANTHANUM-LITHIUM-BINOL COMPLEX: (2S,3S)-2-NITRO-5-PHENYL-1,3-PENTANEDIOL (L-threo-Pentitol, 1,2,4-trideoxy-4-nitro-1-phenyl-) Condensation | Preparation of L-threo-Pentitol, 1,2,4-trideoxy-4-nitro-1-phenyl-. Submitted by Hiroaki Sasai1a , Shizue Watanabe, Takeyuki Suzuki, and Masakatsu Shibasaki1b. DOI: 10.15227/orgsyn.078.0014. | Organic Syntheses | |
| Claisen-Schmidt aldol: benzaldehyde + acetone → benzalacetone Scale Condensation | Textbook aldol condensation. Acetone's α-carbon (deprotonated by aqueous NaOH) attacks benzaldehyde, then β-elimination dehydrates the aldol intermediate to give the E-enone benzalacetone (4-phenyl-3-… | Claisen-Schmidt 1881 | |
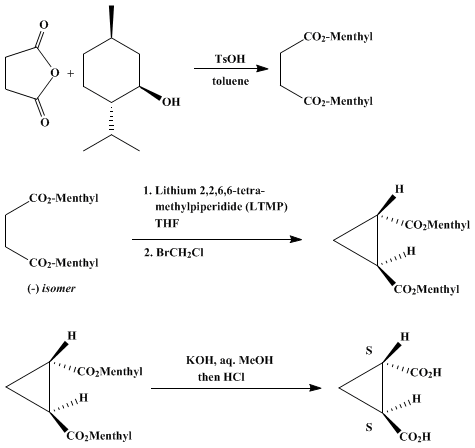
| CONDENSATION OF (−)-DIMENTHYL SUCCINATE DIANION WITH 1,ω-DIHALIDES: (+)-(1S,2S)-CYCLOPROPANE-1,2-DICARBOXYLIC ACID (1,2-Cyclopropanedicarboxylic acid, (1S,2S)-(+)-) Condensation | Preparation of 1,2-Cyclopropanedicarboxylic acid, (1S,2S)-(+)-. Submitted by Kyoji Furuta, Kiyoshi Iwanaga, and Hisashi Yamamoto1. DOI: 10.15227/orgsyn.067.0076. | Organic Syntheses | |
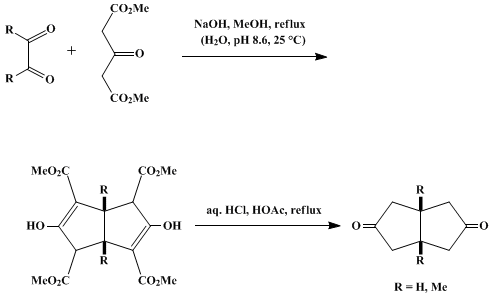
| CONDENSATION OF DIMETHYL 1,3-ACETONEDICARBOXYLATE WITH 1,2-DICARBONYL COMPOUNDS: cis-BICYCLO[3.3.0]OCTANE-3,7-DIONES (2,5(1H,3H)-Pentalenedione, tetrahydro-, cis-, and 2,5(1H,3H)-pentalenedione, tetrahydro-, 3a,6a-dimethyl-, cis) Condensation | Preparation of 2,5(1H,3H)-Pentalenedione, tetrahydro-, cis-, and 2,5(1H,3H)-pentalenedione, tetrahydro-, 3a,6a-dimethyl-, cis. Submitted by Steven H. Bertz1, James M. Cook2, Ali Gawish2, and Ulrich We… | Organic Syntheses | |
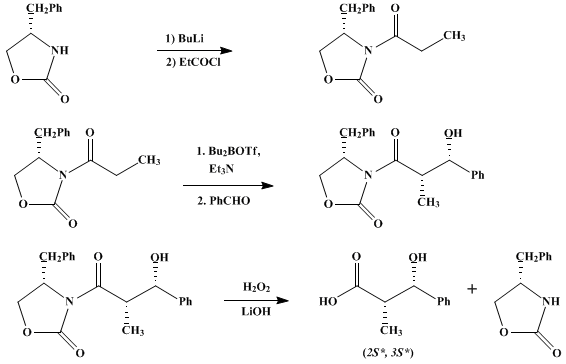
| DIASTEREOSELECTIVE ALDOL CONDENSATION USING A CHIRAL OXAZOLIDINONE AUXILIARY: (2S*,3S*)-3-HYDROXY-3-PHENYL-2-METHYLPROPANOIC ACID (Benzenepropanoic acid, β-hydroxy-α-methyl-, [S-(R*,R*)]-) Condensation | Preparation of Benzenepropanoic acid, β-hydroxy-α-methyl-, [S-(R*,R*)]-. Submitted by James R. Gage and David A. Evans1. DOI: 10.15227/orgsyn.068.0083. | Organic Syntheses | |
| Dieckmann condensation Condensation | The Dieckmann condensation is the intramolecular chemical reaction of diesters with base to give β-keto esters. It is named after the German chemist Walter Dieckmann (1869–1925). The equivalent interm… | Wikipedia: Category:Name reactions | |
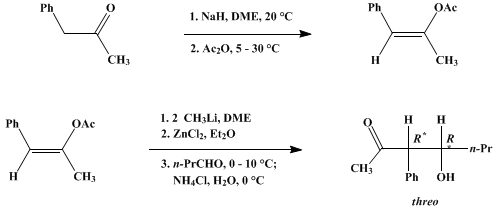
| DIRECTED ALDOL CONDENSATIONS: threo-4-HYDROXY-3-PHENYL-2-HEPTANONE (2-Heptanone, 4-hydroxy-3-phenyl-, (R,R)-) Condensation | Preparation of 2-Heptanone, 4-hydroxy-3-phenyl-, (R,R)-. Submitted by Robert A. Auerbach, David S. Crumrine, David L. Ellison, and Herbert O. House1. DOI: 10.15227/orgsyn.054.0049. | Organic Syntheses | |
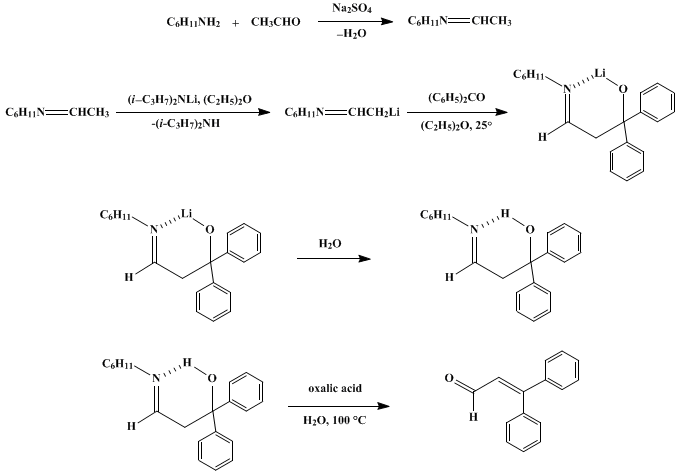
| DIRECTED ALDOL CONDENSATIONS: β-PHENYLCINNAMALDEHYDE (2-Propenal, 3,3-diphenyl-) Condensation | Preparation of 2-Propenal, 3,3-diphenyl-. Submitted by G. Wittig1 and A. Hesse. DOI: 10.15227/orgsyn.050.0066. | Organic Syntheses | |
| Enantioselective Nitroaldol (Henry) Reaction of p-Nitrobenzaldehyde and Nitromethane Using a Copper (II) Complex Derived from (R,R)-1,2-Diaminocyclohexane: (1S)-1-(4-Nitrophenyl)-2-nitroethane-1-ol Condensation | Preparation of Enantioselective Nitroaldol (Henry) Reaction of p-Nitrobenzaldehyde and Nitromethane Using a Copper (II) Complex Derived from (R,R)-1,2-Diaminocyclohexane: (1S)-1-(4-Nitrophenyl)-2-nitr… | Organic Syntheses | |
| Grieco three-component condensation Condensation | The Grieco three-component condensation is an organic chemistry reaction that produces nitrogen-containing six-member heterocycles via a multi-component reaction of an aldehyde, a nitrogen component, … | Wikipedia: Category:Name reactions | |
| Japp–Maitland condensation Condensation | The Japp–Maitland condensation is an organic reaction and a type of Aldol reaction and a tandem reaction. In a reaction between the ketone 3-pentanone and the aldehyde benzaldehyde catalyzed by base t… | Wikipedia: Category:Name reactions | |
| Knoevenagel condensation Condensation | In organic chemistry, the Knoevenagel condensation (pronounced [ˈknøːvənaːɡl̩]) reaction is a type of chemical reaction named after German chemist Emil Knoevenagel. It is a modification of the aldol c… | Wikipedia: Category:Name reactions | |
| Mukaiyama aldol addition Condensation | In organic chemistry, the Mukaiyama aldol addition is an organic reaction and a type of aldol reaction between a silyl enol ether (R2C=CR−O−Si(CH3)3) and an aldehyde (R−CH=O) or formate (R−O−CH=O). Th… | Wikipedia: Category:Name reactions | |
| Mukaiyama hydration Condensation | The Mukaiyama hydration is an organic reaction involving formal addition of an equivalent of water across an olefin by the action of catalytic bis(acetylacetonato)cobalt(II) complex, phenylsilane and … | Wikipedia: Category:Name reactions | |
| ORGANOCATALYTIC ENANTIOSELECTIVE SYNTHESIS OF BICYCLIC ß-LACTONES FROM ALDEHYDE ACIDS VIA NUCLEOPHILE-CATALYZED ALDOL-LACTONIZATION (NCAL) Condensation | Preparation of ORGANOCATALYTIC ENANTIOSELECTIVE SYNTHESIS OF BICYCLIC ß-LACTONES FROM ALDEHYDE ACIDS VIA NUCLEOPHILE-CATALYZED ALDOL-LACTONIZATION (NCAL). Submitted by Henry Nguyen, Seongho Oh, Huda H… | Organic Syntheses | |
| Pechmann condensation Condensation | The Pechmann condensation is a synthesis of coumarins, starting from a phenol and a carboxylic acid or ester containing a β-carbonyl group. The condensation is performed under acidic conditions. The… | Wikipedia: Category:Name reactions | |
| Perkin reaction Condensation | The Perkin reaction is an organic reaction developed by English chemist William Henry Perkin in 1868 that is used to make cinnamic acids. It gives an α,β-unsaturated aromatic acid or α-substituted β-… | Wikipedia: Category:Name reactions | |
| Perkin rearrangement Condensation | The Perkin rearrangement (coumarin–benzofuran ring contraction) is a rearrangement reaction in which a 2-halocoumarin in the presence of hydroxide undergoes a ring contraction to form a benzofuran. Th… | Wikipedia: Category:Name reactions | |
| (Sa,S)-N-[2´-(4-METHYLPHENYLSULFONAMIDO)-1,1´-BINAPHTHYL-2-YL]PYRROLIDINE-2-CARBOXAMIDE: AN ORGANOCATALYST FOR THE DIRECT ALDOL REACTION Condensation | Preparation of (Sa,S)-N-[2´-(4-METHYLPHENYLSULFONAMIDO)-1,1´-BINAPHTHYL-2-YL]PYRROLIDINE-2-CARBOXAMIDE: AN ORGANOCATALYST FOR THE DIRECT ALDOL REACTION. Submitted by Santiago F. Viózquez, 1Gabriela Gu… | Organic Syntheses | |
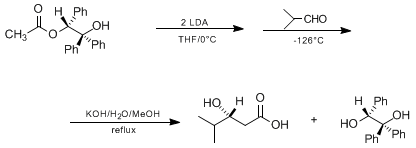
| STEREOSELECTIVE ALDOL REACTION OF DOUBLY DEPROTONATED (R)-(+)-2-HYDROXY-1,2,2-TRIPHENYLETHYL ACETATE (HYTRA): (R)-3-HYDROXY-4-METHYLPENTANOIC ACID (Pentanoic acid, 3-hydroxy-4-methyl-, (R)-) Condensation | Preparation of Pentanoic acid, 3-hydroxy-4-methyl-, (R)-. Submitted by Manfred Braun and Silke Gräf1. DOI: 10.15227/orgsyn.072.0038. | Organic Syntheses | |
| Stobbe condensation Condensation | The Stobbe condensation entails the reaction of an aldehyde or ketone with an ester of succinic acid to generate alkylidene succinic acid or related derivatives. The reaction consumes one equivalent … | Wikipedia: Category:Name reactions | |
| SYNTHESIS AND DIASTEREOSELECTIVE ALDOL REACTIONS OF A THIAZOLIDINETHIONE CHIRAL AUXILIARY Condensation | Preparation of SYNTHESIS AND DIASTEREOSELECTIVE ALDOL REACTIONS OF A THIAZOLIDINETHIONE CHIRAL AUXILIARY. Submitted by Michael T. Crimmins, Hamish S. Christie, and Colin O. Hughes1. DOI: 10.15227/orgs… | Organic Syntheses | |
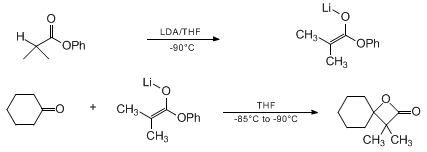
| SYNTHESIS OF β-LACTONES BY ALDOLIZATION OF KETONES WITH PHENYL ESTER ENOLATES: 3,3-DIMETHYL-1-OXASPIRO[3.5]NONAN-2-ONE (1-Oxaspiro[3.5]nonan-2-one, 3,3-dimethyl-) Condensation | Preparation of 1-Oxaspiro[3.5]nonan-2-one, 3,3-dimethyl-. Submitted by Christine Wedler and Hans Schick1. DOI: 10.15227/orgsyn.075.0116. | Organic Syntheses | |
| THE USE OF POLYSTYRYLSULFONYL CHLORIDE RESIN AS A SOLID SUPPORTED CONDENSATION REAGENT FOR THE FORMATION OF ESTERS: SYNTHESIS OF N-[(9-FLUORENYLMETHOXY)CARBONYL]-L-ASPARTIC ACID; α tert-BUTYL ESTER, β-(2-ETHYL[(1E)-(4-NITROPHENYL)AZO]PHENYL]AMINO]ETHYL ESTER Condensation | Preparation of THE USE OF POLYSTYRYLSULFONYL CHLORIDE RESIN AS A SOLID SUPPORTED CONDENSATION REAGENT FOR THE FORMATION OF ESTERS: SYNTHESIS OF N-[(9-FLUORENYLMETHOXY)CARBONYL]-L-ASPARTIC ACID; α tert… | Organic Syntheses | |
| Ullmann condensation Condensation | The Ullmann condensation or Ullmann-type reaction is the copper-promoted conversion of aryl halides to aryl ethers, aryl thioethers, aryl nitriles, and aryl amines. These reactions are examples of cro… | Wikipedia: Category:Name reactions |