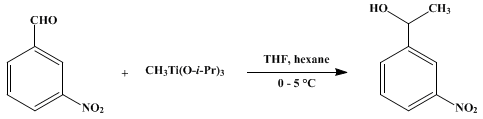Browse · Reactions
Reactions
88 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
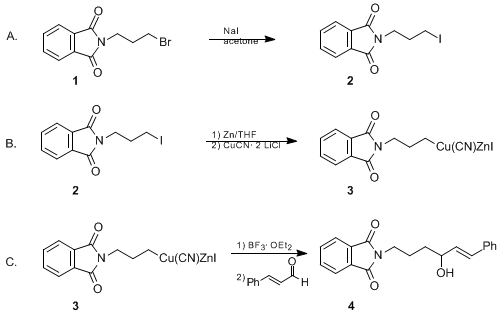
| 1,2-ADDITION OF A FUNCTIONALIZED ZINC-COPPER ORGANOMETALLIC [RCu(CN)ZnI] TO AN α,β-UNSATURATED ALDEHYDE: (E)-2-(4-HYDROXY-6-PHENYL-5-HEXENYL)-1H-ISOINDOLE-1,3(2H)-DIONE (1H-Isoindole-1,3(2H)-dione, 2-(4-hydroxy-6-phenyl-5-hexenyl)-) Addition | Preparation of 1H-Isoindole-1,3(2H)-dione, 2-(4-hydroxy-6-phenyl-5-hexenyl)-. Submitted by Ming Chang P. Yeh, Huai Gu Chen, and Paul Knochel1. DOI: 10.15227/orgsyn.070.0195. | Organic Syntheses | |
| 3'-NITRO-1-PHENYLETHANOL BY ADDITION OF METHYLTRIISOPROPOXYTITANIUM TO m-NITROBENZALDEHYDE (Benzenemethanol, α-methyl-3-nitro-) Addition | Preparation of Benzenemethanol, α-methyl-3-nitro-. Submitted by René Imwinkelried and Dieter Seebach1. DOI: 10.15227/orgsyn.067.0180. | Organic Syntheses | |
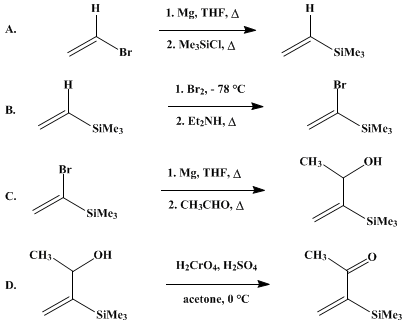
| 3-TRIMETHYLSILYL-3-BUTEN-2-ONE: A MICHAEL ACCEPTOR (3-Buten-2-one, 3-(trimethylsilyl)-) Addition | Preparation of 3-Buten-2-one, 3-(trimethylsilyl)-. Submitted by Robert K. Boeckman, Jr.12, David M. Blum1, Bruce Ganem3, and Neil Halvey3. DOI: 10.15227/orgsyn.058.0152. | Organic Syntheses | |
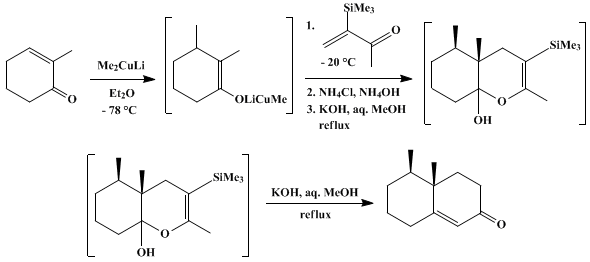
| 3-TRIMETHYLSILYL-3-BUTEN-2-ONE AS MICHAEL ACCEPTOR FOR CONJUGATE ADDITION-ANNELATION: cis-4,4a,5,6,7,8-HEXAHYDRO-4a,5-DIMETHYL-2(3H)-NAPHTHALENONE (2(3H)-Napthalenone, 4,4a,5,6,7,8-hexahydro-, 4a,5-dimethyl-, cis-, (±)-) Addition | Preparation of 2(3H)-Napthalenone, 4,4a,5,6,7,8-hexahydro-, 4a,5-dimethyl-, cis-, (±)-. Submitted by Robert K. Boeckman, Jr.1, David M. Blum, and Bruce Ganem2. DOI: 10.15227/orgsyn.058.0158. | Organic Syntheses | |
| [4+2] CYCLOADDITION OF 1-DIMETHYLAMINO-3-tert-BUTYLDIMETHYLSILOXY-1,3-BUTADIENE WITH METHYL ACRYLATE: 4-HYDROXYMETHYL-2-CYCLOHEXEN-1-ONE (2-Cyclohexen-1-one, 4-(hydroxymethyl)-) Cycloaddition | Preparation of 2-Cyclohexen-1-one, 4-(hydroxymethyl)-. Submitted by Sergey A. Kozmin, Shuwen He, and Viresh H. Rawal1. DOI: 10.15227/orgsyn.078.0160. | Organic Syntheses | |
| [4+3] CYCLOADDITION IN WATER. SYNTHESIS OF 2,4-endo,endo-DIMETHYL-8-OXABICYCLO[3.2.1]OCT-6-EN-3-ONE (8-Oxabicyclo[3.2.1]oct-6-en-3-one, 2,4-dimethyl-, (endo,endo)-) Cycloaddition | Preparation of 8-Oxabicyclo[3.2.1]oct-6-en-3-one, 2,4-dimethyl-, (endo,endo)-. Submitted by Mark Lautens and Giliane Bouchain1. DOI: 10.15227/orgsyn.079.0251. | Organic Syntheses | |
| [4 + 3] CYCLOADDITION OF AMINOALLYL CATIONS WITH 1,3-DIENES: 11-OXATRICYCLO[4.3.1.12,5]UNDEC-3-EN-10-ONE (11-Oxatricyclo[4.3.1.12,5]undec-3-en-10-one,(1α,2β,5β,6α)-) Cycloaddition | Preparation of 11-Oxatricyclo[4.3.1.12,5]undec-3-en-10-one,(1α,2β,5β,6α)-. Submitted by Jonghoon Oh, Chewki Ziani-Cherif, Jong-Ryoo Choi, and Jin Kun Cha1,2. DOI: 10.15227/orgsyn.078.0212. | Organic Syntheses | |
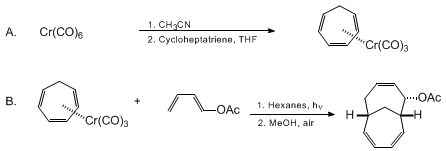
| 7α-ACETOXY-(1Hβ, 6Hβ)-BICYCLO[4.4.1]UNDECA-2,4,8-TRIENE VIA CHROMIUM-MEDIATED HIGHER ORDER CYCLOADDITION (Bicyclo[4.4.1]undeca-3,7,9-triene-2-ol, acetate, endo- (±)-) Cycloaddition | Preparation of Bicyclo[4.4.1]undeca-3,7,9-triene-2-ol, acetate, endo- (±)-. Submitted by James H. Rigby1 and Kevin R. Fales. DOI: 10.15227/orgsyn.077.0121. | Organic Syntheses | |
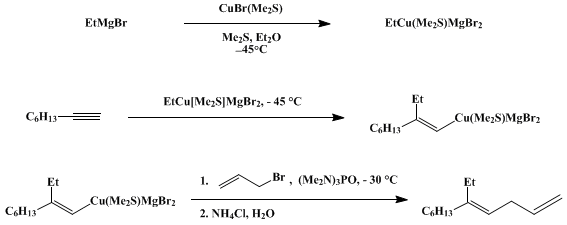
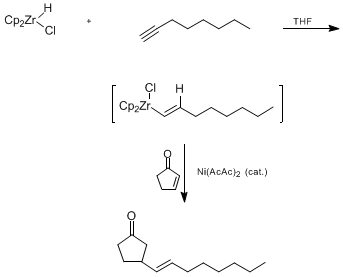
| ADDITION OF AN ETHYLCOPPER COMPLEX TO 1-OCTYNE: (E)-5-ETHYL-1,4-UNDECADIENE (1,4-Undecadiene, 5-ethyl-, (E)-) Addition | Preparation of 1,4-Undecadiene, 5-ethyl-, (E)-. Submitted by Ramnath S. Iyer and Paul Helquist1. DOI: 10.15227/orgsyn.064.0001. | Organic Syntheses | |
| ADDITION OF DIALKYLAMINES TO MYRCENE: N,N-DIETHYLGERANYLAMINE (2,6-Octadien-1-amine, N,N-diethyl-3,7-dimethyl-, (E)-) Addition | Preparation of 2,6-Octadien-1-amine, N,N-diethyl-3,7-dimethyl-, (E)-. Submitted by Kunihiko Takabe1, Takao Katagiri1, Juntaro Tanaka1, Tsutomu Fujita2, Shoji Watanabe2, and Kyoichi Suga2. DOI: 10.1522… | Organic Syntheses | |
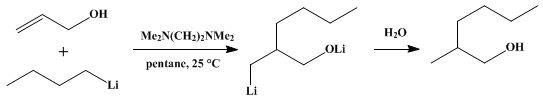
| ADDITION OF ORGANOLITHIUM REAGENTS TO ALLYL ALCOHOL: 2-METHYL-1-HEXANOL (1-Hexanol, 2-methyl-) Addition | Preparation of 1-Hexanol, 2-methyl-. Submitted by J. K. Crandall1 and A. C. Rojas. DOI: 10.15227/orgsyn.055.0001. | Organic Syntheses | |
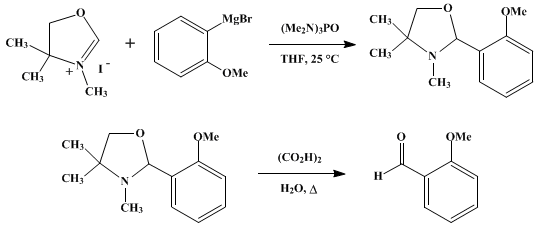
| ALDEHYDES FROM 4,4-DIMETHYL-2-OXAZOLINE AND GRIGNARD REAGENTS: o-ANISALDEHYDE (Benzaldehyde, 2-methoxy-) Organometallic addition | Preparation of Benzaldehyde, 2-methoxy-. Submitted by R. S. Brinkmeyer, E. W. Collington, and A. I. Meyers1. DOI: 10.15227/orgsyn.054.0042. | Organic Syntheses | |
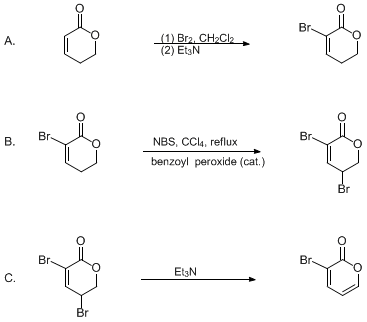
| AN IMPROVED PREPARATION OF 3-BROMO-2H-PYRAN-2-ONE: AN AMBIPHILIC DIENE FOR DIELS-ALDER CYCLOADDITIONS (2H-Pyran-2-one, 3-bromo-) Cycloaddition | Preparation of 2H-Pyran-2-one, 3-bromo-. Submitted by G. H. Posner, K. Afarinkia, and H. Dai1. DOI: 10.15227/orgsyn.073.0231. | Organic Syntheses | |
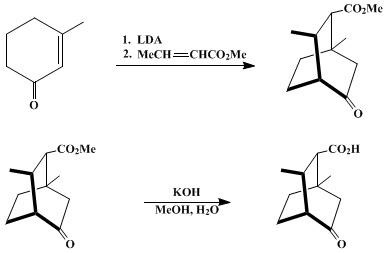
| APROTIC DOUBLE MICHAEL ADDITION: 1,3-DIMETHYL-5-OXOBICYCLO[2.2.2]OCTANE-2-CARBOXYLIC ACID (Bicyclo[2.2.2]octane-2-carboxylic acid, 1,3-dimethyl-5-oxo-) Addition | Preparation of Bicyclo[2.2.2]octane-2-carboxylic acid, 1,3-dimethyl-5-oxo-. Submitted by Dietrich Spitzner and Anita Engler1. DOI: 10.15227/orgsyn.066.0037. | Organic Syntheses | |
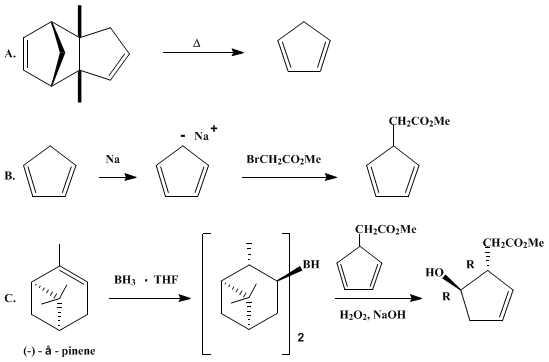
| ASYMMETRIC HYDROBORATION OF 5-SUBSTITUTED CYCLOPENTADIENES: SYNTHESIS OF METHYL (1 R,5R)-5-HYDROXY-2-CYCLOPENTENE-1-ACETATE (2-Cyclopentene-1-acetic acid, 5-hydroxy-, methyl ester, (1R-trans)-) Addition | Preparation of 2-Cyclopentene-1-acetic acid, 5-hydroxy-, methyl ester, (1R-trans)-. Submitted by John J. Partridge, Naresh K. Chadha, and Milan R. Uskokovic1. DOI: 10.15227/orgsyn.063.0044. | Organic Syntheses | |
| Azide-alkyne Huisgen cycloaddition Cycloaddition | The azide-alkyne Huisgen cycloaddition is a 1,3-dipolar cycloaddition between an azide and a terminal or internal alkyne to give a 1,2,3-triazole. Rolf Huisgen was the first to understand the scope of… | Wikipedia: Category:Name reactions | |
| Barbier reaction Organometallic addition | The Barbier reaction is an organometallic reaction between an alkyl halide (chloride, bromide, iodide), a carbonyl group and a metal. The reaction can be performed using magnesium, aluminium, zinc, in… | Wikipedia: Category:Name reactions | |
| Barbier–Wieland degradation Organometallic addition | The Barbier–Wieland degradation is a procedure for shortening the carbon chain of a carboxylic acid by one carbon. It only works when the carbon adjacent to the carboxyl is a simple methylene bridge (… | Wikipedia: Category:Name reactions | |
| BENZYL ISOPROPOXYMETHYL CARBAMATE - AN AMINOMETHYLATING REAGENT FOR MANNICH REACTIONS OF TITANIUM ENOLATES Addition | Preparation of BENZYL ISOPROPOXYMETHYL CARBAMATE - AN AMINOMETHYLATING REAGENT FOR MANNICH REACTIONS OF TITANIUM ENOLATES. Submitted by Hartmut Meyer1, Albert K. Beck2, Radovan Sebesta2, and Dieter Se… | Organic Syntheses | |
| Bradsher cycloaddition Cycloaddition | The Bradsher cycloaddition reaction, also known as the Bradsher cyclization reaction is a form of the Diels–Alder reaction which involves the [4+2] addition of a common dienophile with a cationic aro… | Wikipedia: Category:Name reactions | |
| CATALYTIC ASYMMETRIC ADDITION OF AN IN-SITU PREPARED ARYLZINC TO CYCLOHEXANECARBOXALDEHYDE: (R)-(+)-α-CYCLOHEXYL-3-METHOXY-BENZENEMETHANOL Addition | Preparation of CATALYTIC ASYMMETRIC ADDITION OF AN IN-SITU PREPARED ARYLZINC TO CYCLOHEXANECARBOXALDEHYDE: (R)-(+)-α-CYCLOHEXYL-3-METHOXY-BENZENEMETHANOL. Submitted by Albert M. DeBerardinis, Mark Tur… | Organic Syntheses | |
| CATALYTIC ENANTIOSELECTIVE ADDITION OF DIALKYLZINCS TO ALDEHYDES USING (2S)-(−)-3-exo-(DIMETHYLAMINO)ISOBORNEOL [(2S)-DAIB]: (S)-1-PHENYL-1-PROPANOL (Benzenemethanol, α-ethyl-, (S)-) Addition | Preparation of Benzenemethanol, α-ethyl-, (S)-. Submitted by Masato Kitamura, Hiromasa Oka, Seiji Suga, and Ryoji Noyori1. DOI: 10.15227/orgsyn.079.0139. | Organic Syntheses | |
| CATALYTIC ENANTIOSELECTIVE ADDITION OF TERMINAL ALKYNES TO ALDEHYDES: PREPARATION OF (S)-(-)-1,3-DIPHENYL-2-PROPYN-1-OL AND (S)-(-)-4-METHYL-1-PHENYL-2-PENTYN-1,4-DIOL Addition | Preparation of CATALYTIC ENANTIOSELECTIVE ADDITION OF TERMINAL ALKYNES TO ALDEHYDES: PREPARATION OF (S)-(-)-1,3-DIPHENYL-2-PROPYN-1-OL AND (S)-(-)-4-METHYL-1-PHENYL-2-PENTYN-1,4-DIOL. Submitted by Ryo… | Organic Syntheses | |
| CONJUGATE ADDITION OF A VINYLZIRCONIUM REAGENT: 3-(1-OCTEN-1-YL)CYCLOPENTANONE (Cyclopentanone, 3-(1-octenyl)-, (E)-) Addition | Preparation of Cyclopentanone, 3-(1-octenyl)-, (E)-. Submitted by Ruen Chu Sun1, Masami Okabe1, David L. Coffen1, and Jeffrey Schwartz2. DOI: 10.15227/orgsyn.071.0083. | Organic Syntheses | |
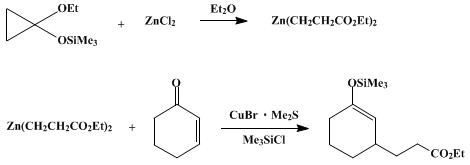
| COPPER-CATALYZED CONJUGATE ADDITION OF A ZINC HOMOENOLATE: ETHYL 3-[3-(TRIMETHYLSILYLOXY)CYCLOHEX-2-ENYL]PROPIONATE (2-Cyclohexene-1-propanoic acid, 3-[(trimethylsilyl)oxy]-, ethyl ester) Addition | Preparation of 2-Cyclohexene-1-propanoic acid, 3-[(trimethylsilyl)oxy]-, ethyl ester. Submitted by Eiichi Nakamura and Isao Kuwajima1. DOI: 10.15227/orgsyn.066.0043. | Organic Syntheses | |
| COPPER-CATALYZED CONJUGATE ADDITION OF FUNCTIONALIZED ORGANOZINC REAGENTS TO α,β-UNSATURATED KETONES: ETHYL 5-(3-OXOCYCLOHEXYL)PENTANOATE (Pentanoic acid, 5-(3-oxocyclohexyl)-, ethyl ester) Addition | Preparation of Pentanoic acid, 5-(3-oxocyclohexyl)-, ethyl ester. Submitted by B. H. Lipshutz1 , M. R. Wood, and R. Tirado. DOI: 10.15227/orgsyn.076.0252. | Organic Syntheses | |
| COPPER(I)-CATALYZED PHOTOCYCLOADDITION: 3,3-DIMETHYL-cis-BICYCLO[3.2.0]HEPTAN-2-ONE (Bicyclo[3.2.0]heptan-2-one, 3,3-dimethyl-) Cycloaddition | Preparation of Bicyclo[3.2.0]heptan-2-one, 3,3-dimethyl-. Submitted by Robert G. Salomon and Subrata Ghosh1. DOI: 10.15227/orgsyn.062.0125. | Organic Syntheses | |
| COPPER(I) IODIDE DIMETHYL SULFIDE CATALYZED ADDITION OF A VINYLZIRCONIUM REAGENT (PREPARATION OF 4-PHENYL-5(E)-DECEN-2-ONE) Addition | Preparation of PREPARATION OF 4-PHENYL-5(E)-DECEN-2-ONE. Submitted by Amer El-Batta and Mikael Bergdahl1. DOI: 10.15227/orgsyn.084.0192. | Organic Syntheses | |
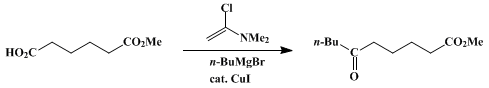
| Cross-Coupling of Alkenyl/Aryl Carboxylates with Grignard Reagents via Fe-Catalyzed C-O Bond Activation Organometallic addition | Preparation of Cross-Coupling of Alkenyl/Aryl Carboxylates with Grignard Reagents via Fe-Catalyzed C-O Bond Activation. Submitted by Bi-Jie Li, 1 Xi-Sha Zhang1 and Zhang-Jie Shi1*. DOI: 10.15227/orgsy… | Organic Syntheses | |
| Cu-CATALYZED AZIDE-ALKYNE CYCLOADDITION: PREPARATION OF TRIS((1-BENZYL-1H-1,2,3-TRIAZOLYL)METHYL)AMINE Cycloaddition | Preparation of Cu-CATALYZED AZIDE-ALKYNE CYCLOADDITION: PREPARATION OF TRIS((1-BENZYL-1H-1,2,3-TRIAZOLYL)METHYL)AMINE. Submitted by Jason E. Hein, Larissa B. Krasnova, Masayuki Iwasaki, and Valery V.F… | Organic Syntheses | |
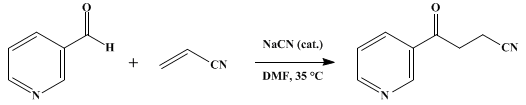
| CYANIDE-CATALYZED CONJUGATE ADDITION OF ARYL ALDEHYDES: 4-OXO-4-(3-PYRIDYL)BUTYRONITRILE (3-Pyridinebutanenitrile, γ-oxo) Addition | Preparation of 3-Pyridinebutanenitrile, γ-oxo. Submitted by H. Stetter1, H. Kuhlmann, and G. Lorenz. DOI: 10.15227/orgsyn.059.0053. | Organic Syntheses | |
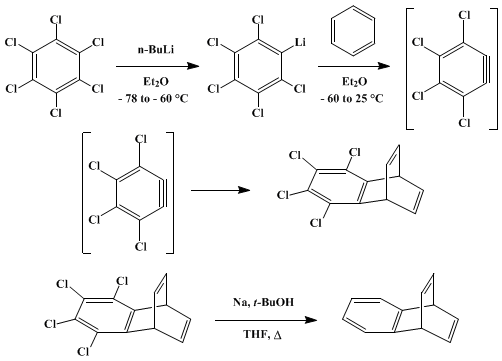
| DIELS-ALDER ADDITION OF PERCHLOROBENZYNE: BENZOBARRELENE (1,4-Ethenonaphthalene, 1,4-dihydro) Addition | Preparation of 1,4-Ethenonaphthalene, 1,4-dihydro. Submitted by Neil J. Hales, Harry Heaney1, John H. Hollinshead, and Pritpal Singh. DOI: 10.15227/orgsyn.059.0071. | Organic Syntheses | |
| Diels–Alder cycloaddition: cyclopentadiene + maleic anhydride → endo-norbornene-2,3-dicarboxylic anhydride Scale Cycloaddition | Textbook [4+2] cycloaddition. Electron-rich diene (cyclopentadiene, freshly cracked from its dimer) reacts with electron-poor dienophile (maleic anhydride) in a concerted, suprafacial, endo-selective … | Diels-Alder 1928 | |
| DIETHYLAMINOTRIMETHYLSILANE-CATALYZED 1,4-ADDITION OF ALDEHYDES TO VINYL KETONES: (3R)-3,7-DIMETHYL-2-(3-OXOBUTYL)-6-OCTENAL ((6-Octenal, 3,7-dimethyl-2-(3-oxobutyl)-, (3R)-)) Addition | Preparation of (6-Octenal, 3,7-dimethyl-2-(3-oxobutyl)-, (3R)-). Submitted by Hisahiro Hagiwara, Hiroki Ono, and Takashi Hoshi.1. DOI: 10.15227/orgsyn.080.0195. | Organic Syntheses | |
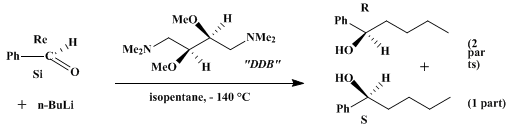
| ENANTIOSELECTIVE ADDITION OF BUTYLLITHIUM IN THE PRESENCE OF THE CHIRAL COSOLVENT DDB: (R)-( + )-1-PHENYL-1-PENTANOL (Benzenemethanol, α-butyl-, (R)-) Addition | Preparation of Benzenemethanol, α-butyl-, (R)-. Submitted by Dieter Seebach and August Hidber1. DOI: 10.15227/orgsyn.061.0042. | Organic Syntheses | |
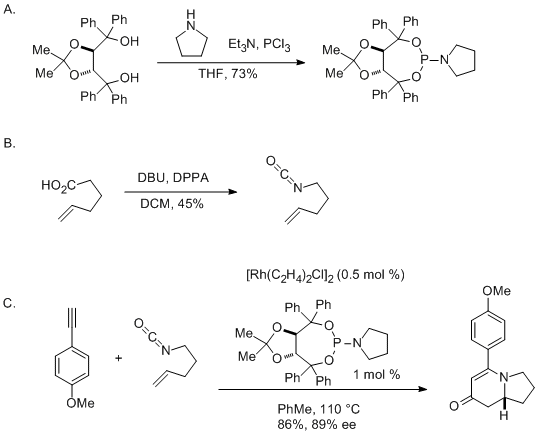
| Enantioselective Rhodium-Catalyzed [2+2+2] Cycloaddition of Pentenyl Isocyanate and 4-Ethynylanisole: Preparation and Use of Taddol-pyrrolidine Phosphoramidite Cycloaddition | Preparation of Enantioselective Rhodium-Catalyzed [2+2+2] Cycloaddition of Pentenyl Isocyanate and 4-Ethynylanisole: Preparation and Use of Taddol-pyrrolidine Phosphoramidite. Submitted by Kevin M. Ob… | Organic Syntheses | |
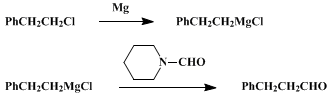
| FORMYL TRANSFER TO GRIGNARD REAGENTS WITH N-FORMYLPIPERIDINE: 3-PHENYLPROPIONALDEHYDE Organometallic addition | Preparation of FORMYL TRANSFER TO GRIGNARD REAGENTS WITH N-FORMYLPIPERIDINE: 3-PHENYLPROPIONALDEHYDE. Submitted by George A. Olah and Massoud Arvanaghi1. DOI: 10.15227/orgsyn.064.0114. | Organic Syntheses | |
| GENERATION AND [2+2] CYCLOADDITIONS OF THIO-SUBSTITUTED KETENES: trans-1-(4-METHOXYPHENYL)-4-PHENYL-3-(PHENYLTHIO)AZETIDIN-2-ONE ((2-Azetidinone, 1-(4-methoxyphenyl)-4-phenyl-3-(phenylthio)-, trans-)) Cycloaddition | Preparation of (2-Azetidinone, 1-(4-methoxyphenyl)-4-phenyl-3-(phenylthio)-, trans-). Submitted by Rick L. Danheiser, Iwao Okamoto, Michael D. Lawlor, and Thomas W. Lee.1. DOI: 10.15227/orgsyn.080.016… | Organic Syntheses | |
| Grignard addition: phenylmagnesium bromide + benzaldehyde → diphenylmethanol Scale Organometallic addition | Textbook Grignard addition. Phenylmagnesium bromide (PhMgBr) attacks benzaldehyde's carbonyl carbon; aqueous acidic workup releases diphenylmethanol (benzhydrol). PhMgBr is bought as a 1 M or 3 M solu… | Grignard 1900 | |
| Grignard reaction Organometallic addition | The Grignard reaction (French: [ɡʁiɲaʁ]) is an organometallic chemical reaction in which, according to the classical definition, carbon alkyl, allyl, vinyl, or aryl magnesium halides (Grignard reagent… | Wikipedia: Category:Name reactions | |
| Henry reaction (nitroaldol addition) Scale Addition | Base-catalysed addition of a nitroalkane to an aldehyde or ketone to give a β-nitro alcohol. Key C-C bond-forming step in the Knoevenagel-Henry route to β-nitrostyrenes (e.g. P2NP). | Henry 1895 | |
| HIGHLY EFFICIENT ADDITION OF ACTIVATED METHYLENE COMPOUNDS TO ALKENES CATALYZED BY GOLD AND SILVER Addition | Preparation of HIGHLY EFFICIENT ADDITION OF ACTIVATED METHYLENE COMPOUNDS TO ALKENES CATALYZED BY GOLD AND SILVER. Submitted by Chao-Jun Li1 and Xiaoquan Yao. DOI: 10.15227/orgsyn.084.0222. | Organic Syntheses | |
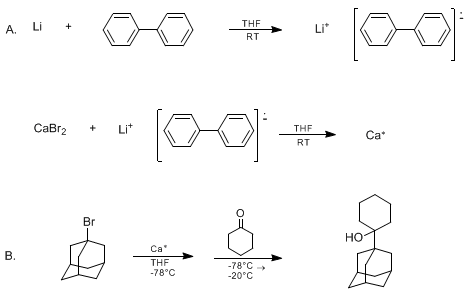
| HIGHLY REACTIVE CALCIUM FOR THE PREPARATION OF ORGANOCALCIUM REAGENTS: 1-ADAMANTYL CALCIUM HALIDES AND THEIR ADDITION TO KETONES: 1-(1-ADAMANTYL)CYCLOHEXANOL (Cyclohexanol, 1-tricyclo[3.3.1.13,7]dec-1-yl) Addition | Preparation of Cyclohexanol, 1-tricyclo[3.3.1.13,7]dec-1-yl. Submitted by Reuben D. Rieke, Tse-Chong Wu, and Loretta I. Rieke1. DOI: 10.15227/orgsyn.072.0147. | Organic Syntheses | |
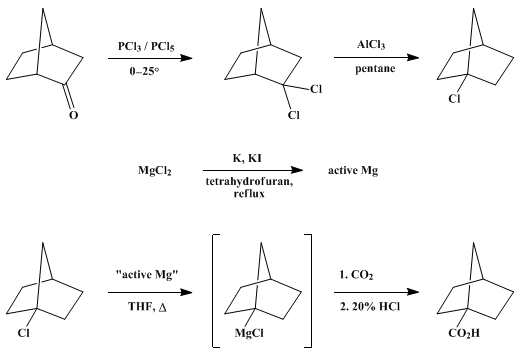
| HIGHLY REACTIVE MAGNESIUM FOR THE PREPARATION OF GRIGNARD REAGENTS: 1-NORBORNANECARBOXYLIC ACID (Bicyclo[2.2.1]heptane-1-carboxylic acid) Organometallic addition | Preparation of Bicyclo[2.2.1]heptane-1-carboxylic acid. Submitted by Reuben D. Rieke1, Stephen E. Bales, Phillip M. Hudnall, Timothy P. Burns, and Graham S. Poindexter. DOI: 10.15227/orgsyn.059.0085. | Organic Syntheses | |
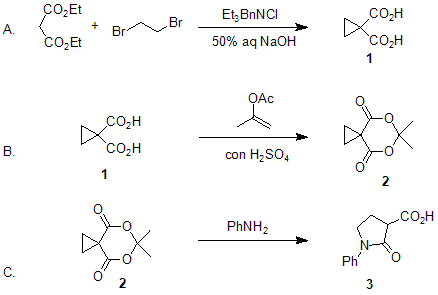
| HOMOCONJUGATE ADDITION OF NUCLEOPHILES TO CYCLOPROPANE-1,1-DICARBOXYLATE DERIVATIVES: 2-OXO-1-PHENYL-3-PYRROLIDINECARBOXYLIC ACID (3-Pyrrolidinecarboxylic acid, 2-oxo-1-phenyl-) Addition | Preparation of 3-Pyrrolidinecarboxylic acid, 2-oxo-1-phenyl-. Submitted by Rajendra K. Singh and Samuel Danishefsky1. DOI: 10.15227/orgsyn.060.0066. | Organic Syntheses | |
| Intramolecular Diels–Alder cycloaddition Cycloaddition | In organic chemistry, an intramolecular Diels-Alder cycloaddition is a Diels–Alder reaction in which the diene and the dienophile are both part of the same molecule. The reaction leads to the formatio… | Wikipedia: Category:Name reactions | |
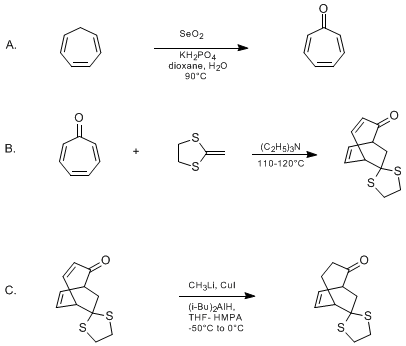
| INVERSE ELECTRON-DEMAND DIELS-ALDER CYCLOADDITION OF A KETENE DITHIOACETAL. COPPER HYDRIDE-PROMOTED REDUCTION OF A CONJUGATED ENONE. 9-DITHIOLANOBICYCLO[3.2.2]NON-6-EN-2-ONE FROM TROPONE Cycloaddition | Preparation of INVERSE ELECTRON-DEMAND DIELS-ALDER CYCLOADDITION OF A KETENE DITHIOACETAL. COPPER HYDRIDE-PROMOTED REDUCTION OF A CONJUGATED ENONE. 9-DITHIOLANOBICYCLO[3.2.2]NON-6-EN-2-ONE FROM TROPON… | Organic Syntheses | |
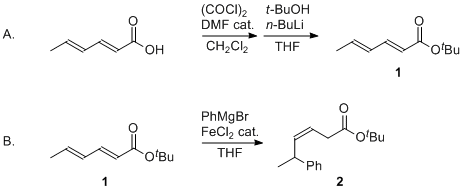
| Iron-Catalyzed Selective Conjugate Addition of Aryl Grignard Reagents to 2,4-Alkadienoates: tert-Butyl (Z)-5-Phenyl-3-hexenoate Organometallic addition | Preparation of Iron-Catalyzed Selective Conjugate Addition of Aryl Grignard Reagents to 2,4-Alkadienoates: tert-Butyl (Z)-5-Phenyl-3-hexenoate. Submitted by Takeshi Hata, Hideyuki Goto, Tomofumi Yokom… | Organic Syntheses | |
| KETONES FROM CARBOXYLIC ACIDS AND GRIGNARD REAGENTS: METHYL 6-OXODECANOATE (Decanoic acid, 6-oxo-, methyl ester) Organometallic addition | Preparation of Decanoic acid, 6-oxo-, methyl ester. Submitted by Tamotsu Fujisawa and Toshio Sato1. DOI: 10.15227/orgsyn.066.0116. | Organic Syntheses | |
| Kharasch addition Addition | The Kharasch addition is an organic reaction and a metal-catalysed free radical addition of CXCl3 compounds (X = Cl, Br, H) to alkenes. The reaction is used to append trichloromethyl or dichloromethy… | Wikipedia: Category:Name reactions |