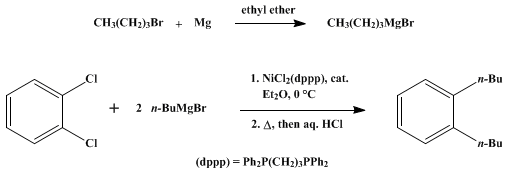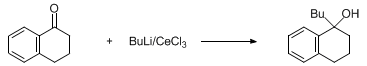Browse · Reactions
Reactions
16 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
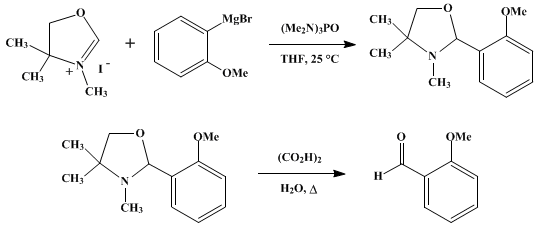
| ALDEHYDES FROM 4,4-DIMETHYL-2-OXAZOLINE AND GRIGNARD REAGENTS: o-ANISALDEHYDE (Benzaldehyde, 2-methoxy-) Organometallic addition | Preparation of Benzaldehyde, 2-methoxy-. Submitted by R. S. Brinkmeyer, E. W. Collington, and A. I. Meyers1. DOI: 10.15227/orgsyn.054.0042. | Organic Syntheses | |
| Barbier reaction Organometallic addition | The Barbier reaction is an organometallic reaction between an alkyl halide (chloride, bromide, iodide), a carbonyl group and a metal. The reaction can be performed using magnesium, aluminium, zinc, in… | Wikipedia: Category:Name reactions | |
| Barbier–Wieland degradation Organometallic addition | The Barbier–Wieland degradation is a procedure for shortening the carbon chain of a carboxylic acid by one carbon. It only works when the carbon adjacent to the carboxyl is a simple methylene bridge (… | Wikipedia: Category:Name reactions | |
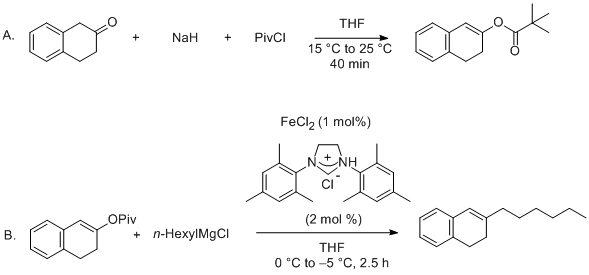
| Cross-Coupling of Alkenyl/Aryl Carboxylates with Grignard Reagents via Fe-Catalyzed C-O Bond Activation Organometallic addition | Preparation of Cross-Coupling of Alkenyl/Aryl Carboxylates with Grignard Reagents via Fe-Catalyzed C-O Bond Activation. Submitted by Bi-Jie Li, 1 Xi-Sha Zhang1 and Zhang-Jie Shi1*. DOI: 10.15227/orgsy… | Organic Syntheses | |
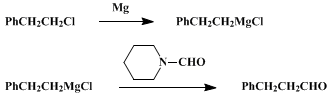
| FORMYL TRANSFER TO GRIGNARD REAGENTS WITH N-FORMYLPIPERIDINE: 3-PHENYLPROPIONALDEHYDE Organometallic addition | Preparation of FORMYL TRANSFER TO GRIGNARD REAGENTS WITH N-FORMYLPIPERIDINE: 3-PHENYLPROPIONALDEHYDE. Submitted by George A. Olah and Massoud Arvanaghi1. DOI: 10.15227/orgsyn.064.0114. | Organic Syntheses | |
| Grignard addition: phenylmagnesium bromide + benzaldehyde → diphenylmethanol Scale Organometallic addition | Textbook Grignard addition. Phenylmagnesium bromide (PhMgBr) attacks benzaldehyde's carbonyl carbon; aqueous acidic workup releases diphenylmethanol (benzhydrol). PhMgBr is bought as a 1 M or 3 M solu… | Grignard 1900 | |
| Grignard reaction Organometallic addition | The Grignard reaction (French: [ɡʁiɲaʁ]) is an organometallic chemical reaction in which, according to the classical definition, carbon alkyl, allyl, vinyl, or aryl magnesium halides (Grignard reagent… | Wikipedia: Category:Name reactions | |
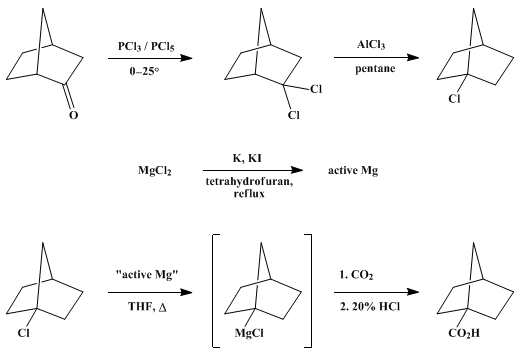
| HIGHLY REACTIVE MAGNESIUM FOR THE PREPARATION OF GRIGNARD REAGENTS: 1-NORBORNANECARBOXYLIC ACID (Bicyclo[2.2.1]heptane-1-carboxylic acid) Organometallic addition | Preparation of Bicyclo[2.2.1]heptane-1-carboxylic acid. Submitted by Reuben D. Rieke1, Stephen E. Bales, Phillip M. Hudnall, Timothy P. Burns, and Graham S. Poindexter. DOI: 10.15227/orgsyn.059.0085. | Organic Syntheses | |
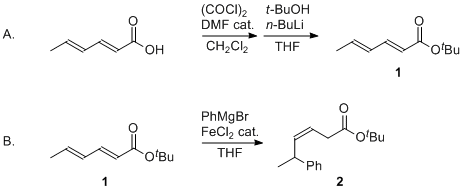
| Iron-Catalyzed Selective Conjugate Addition of Aryl Grignard Reagents to 2,4-Alkadienoates: tert-Butyl (Z)-5-Phenyl-3-hexenoate Organometallic addition | Preparation of Iron-Catalyzed Selective Conjugate Addition of Aryl Grignard Reagents to 2,4-Alkadienoates: tert-Butyl (Z)-5-Phenyl-3-hexenoate. Submitted by Takeshi Hata, Hideyuki Goto, Tomofumi Yokom… | Organic Syntheses | |
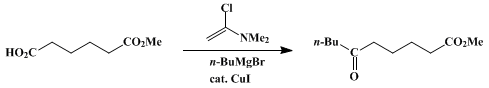
| KETONES FROM CARBOXYLIC ACIDS AND GRIGNARD REAGENTS: METHYL 6-OXODECANOATE (Decanoic acid, 6-oxo-, methyl ester) Organometallic addition | Preparation of Decanoic acid, 6-oxo-, methyl ester. Submitted by Tamotsu Fujisawa and Toshio Sato1. DOI: 10.15227/orgsyn.066.0116. | Organic Syntheses | |
| Lumière–Barbier method Organometallic addition | The Lumière–Barbier method is a method of acetylating aromatic amines in aqueous solutions. Illustrative is the acetylation of aniline. First aniline is dissolved in water using one equivalent of hyd… | Wikipedia: Category:Name reactions | |
| NICKEL-CATALYZED COUPLING OF ARYL O-CARBAMATES WITH GRIGNARD REAGENTS: 2,7-DIMETHYLNAPHTHALENE (Naphthalene, 2,7-dimethyl-) Organometallic addition | Preparation of Naphthalene, 2,7-dimethyl-. Submitted by Carol Dallaire1a , Isabelle Kolber 1b , and Marc Gingras 1c. DOI: 10.15227/orgsyn.078.0042. | Organic Syntheses | |
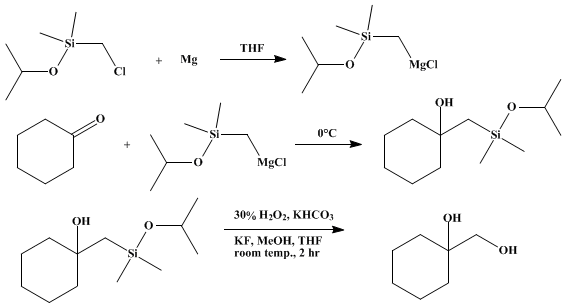
| NUCLEOPHILIC HYDROXYMETHYLATION BY THE (ISOPROPOXYDIMETHYLSILYL)METHYL GRIGNARD REAGENT: 1-(HYDROXYMETHYL)CYCLOHEXANOL FROM CYCLOHEXANONE (Cyclohexanemethanol, 1-hydroxy-) Organometallic addition | Preparation of Cyclohexanemethanol, 1-hydroxy-. Submitted by Kohei Tamao, Neyoshi Ishida, Yoshihiko Ito, and Makoto Kumada1. DOI: 10.15227/orgsyn.069.0096. | Organic Syntheses | |
| PHOSPHINE–NICKEL COMPLEX CATALYZED CROSS-COUPLING OF GRIGNARD REAGENTS WITH ARYL AND ALKENYL HALIDES: 1,2-DIBUTYLBENZENE (Benzene, 1,2-di-n-butyl-) Organometallic addition | Preparation of Benzene, 1,2-di-n-butyl-. Submitted by Makoto Kumada, Kohei Tamao, and Koji Sumitani1. DOI: 10.15227/orgsyn.058.0127. | Organic Syntheses | |
| Reformatsky reaction Organometallic addition | The Reformatsky reaction (sometimes transliterated as Reformatskii reaction) is an organic reaction which condenses aldehydes or ketones with α-halo esters using metallic zinc to form β-hydroxy-esters… | Wikipedia: Category:Name reactions | |
| USE OFCERIUM(III) CHLORIDE IN THE REACTIONS OF CARBONYL COMPOUNDS WITH ORGANOLITHIUMS OR GRIGNARD REAGENTS FOR THE SUPPRESSION OF ABNORMAL REACTIONS:1-BUTYL-1,2,3,4-TETRAHYDRO-1-NAPHTHOL Organometallic addition | Preparation of USE OFCERIUM(III) CHLORIDE IN THE REACTIONS OF CARBONYL COMPOUNDS WITH ORGANOLITHIUMS OR GRIGNARD REAGENTS FOR THE SUPPRESSION OF ABNORMAL REACTIONS:1-BUTYL-1,2,3,4-TETRAHYDRO-1-NAPHTHO… | Organic Syntheses |