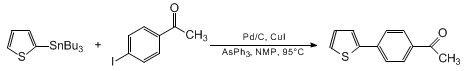Browse · Reactions
Reactions
64 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
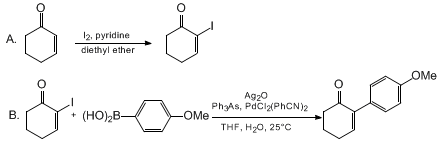
| 2-(4-METHOXYPHENYL)-2-CYCLOHEXEN-1-ONE: PREPARATION OF 2-IODO-2-CYCLOHEXEN-1-ONE AND SUZUKI COUPLING WITH 4-METHOXYPHENYLBORONIC ACID (2-Cyclohexen-1-one, 2-(4-methoxyphenyl)- and 2-Cyclohexen-1-one, 2-iodo-)) Cross-coupling | Preparation of 2-Cyclohexen-1-one, 2-(4-methoxyphenyl)- and 2-Cyclohexen-1-one, 2-iodo-). Submitted by Frederic S. Ruel, Matthew P. Braun, and Carl R. Johnson1. DOI: 10.15227/orgsyn.075.0069. | Organic Syntheses | |
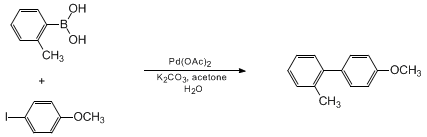
| ACCELERATED SUZUKI COUPLING VIA A LIGANDLESS PALLADIUM CATALYST: 4-METHOXY-2'-METHYLBIPHENYL (1,1'-Biphenyl, 4'-methoxy-2-methyl-) Cross-coupling | Preparation of 1,1'-Biphenyl, 4'-methoxy-2-methyl-. Submitted by Felix E. Goodson, Thomas I. Wallow1 , and Bruce M. Novak2. DOI: 10.15227/orgsyn.075.0061. | Organic Syntheses | |
| ALKYNYL(PHENYL)IODONIUM TOSYLATES: PREPARATION AND STEREOSPECIFIC COUPLING WITH VINYLCOPPER REAGENTS. FORMATION OF CONJUGATED ENYNES. 1-HEXYNYL(PHENYL)IODONIUM TOSYLATE AND (E)-5-PHENYLDODEC-5-EN-7-YNE (Iodine, 1-hexynyl(4-methylbenzenesulfonato-O)phenyl- and Benzene, (1-butyl-1-octen-3-ynyl)-, (E)-) Cross-coupling | Preparation of Iodine, 1-hexynyl(4-methylbenzenesulfonato-O)phenyl- and Benzene, (1-butyl-1-octen-3-ynyl)-, (E)-. Submitted by Peter J. Stang and Tsugio Kitamura1. DOI: 10.15227/orgsyn.070.0215. | Organic Syntheses | |
| B-PROTECTED HALOBORONIC ACIDS FOR ITERATIVE CROSS-COUPLING Cross-coupling | Preparation of B-PROTECTED HALOBORONIC ACIDS FOR ITERATIVE CROSS-COUPLING. Submitted by Steven G. Ballmer, Eric P. Gillis, and Martin D. Burke1. DOI: 10.15227/orgsyn.086.0344. | Organic Syntheses | |
| Buchwald–Hartwig amination Cross-coupling | In organic chemistry, the Buchwald–Hartwig amination is a chemical reaction for the synthesis of carbon–nitrogen bonds via the palladium-catalyzed coupling reactions of amines with aryl halides. Altho… | Wikipedia: Category:Name reactions | |
| Cadiot–Chodkiewicz coupling Cross-coupling | The Cadiot–Chodkiewicz coupling in organic chemistry is a coupling reaction between a terminal alkyne and a haloalkyne catalyzed by a copper(I) salt such as copper(I) bromide and an amine base. The re… | Wikipedia: Category:Name reactions | |
| Castro–Stephens coupling Cross-coupling | The Castro–Stephens coupling is a cross coupling reaction between a copper(I) acetylide and an aryl halide in pyridine, forming a disubstituted alkyne and a copper(I) halide. | Wikipedia: Category:Name reactions | |
| Chan–Lam coupling Cross-coupling | The Chan–Lam coupling reaction, also known as the Chan–Evans–Lam coupling, is a cross-coupling reaction between an aryl boronic acid and an alcohol or an amine to form the corresponding secondary aryl… | Wikipedia: Category:Name reactions | |
| COUPLING OF o-TOLIDINE AND CHICAGO ACID (Preparation of a Salt-Free Azo Dye) Cross-coupling | Preparation of Preparation of a Salt-Free Azo Dye. Submitted by J. L. Hartwell and Louis F. Fieser. DOI: 10.15227/orgsyn.016.0012. | Organic Syntheses | |
| Discussion addendum for: 4-METHOXY-4'-NITROPHENYL. RECENT ADVANCES IN THE STILLE BIARYL COUPLING REACTION AND APPLICATIONS IN COMPLEX NATURAL PRODUCTS SYNTHESIS Cross-coupling | Preparation of Discussion addendum for: 4-METHOXY-4'-NITROPHENYL. RECENT ADVANCES IN THE STILLE BIARYL COUPLING REACTION AND APPLICATIONS IN COMPLEX NATURAL PRODUCTS SYNTHESIS. Submitted by Robert M. … | Organic Syntheses | |
| Discussion Addendum for: PALLADIUM CATALYZED CROSS-COUPLING OF (Z)-1-HEPTENYLDIMETHYLSILANOL WITH 4-IODOANISOLE: (Z)-(1-HEPTENYL)-4-METHOXYBENZENE Cross-coupling | Preparation of Discussion Addendum for: PALLADIUM CATALYZED CROSS-COUPLING OF (Z)-1-HEPTENYLDIMETHYLSILANOL WITH 4-IODOANISOLE: (Z)-(1-HEPTENYL)-4-METHOXYBENZENE. Submitted by Scott E. Denmark * and J… | Organic Syntheses | |
| Discussion Addendum for: Suzuki-Miyaura Cross-Coupling: Preparation of 2'-Vinylacetanilide Cross-coupling | Preparation of Discussion Addendum for: Suzuki-Miyaura Cross-Coupling: Preparation of 2'-Vinylacetanilide. Submitted by Donal F. O'Shea.*1. DOI: 10.15227/orgsyn.089.0202. | Organic Syntheses | |
| Discussion Addendum for: Synthesis of 4-, 5-, and 6-Methyl-2,2'-Bipyridine by a Negishi Cross-Coupling Strategy: 5-Methyl-2,2'-Bipyridine Cross-coupling | Preparation of Discussion Addendum for: Synthesis of 4-, 5-, and 6-Methyl-2,2'-Bipyridine by a Negishi Cross-Coupling Strategy: 5-Methyl-2,2'-Bipyridine. Submitted by Tiandong Liu and Cassandra L. Fra… | Organic Syntheses | |
| Fukuyama coupling Cross-coupling | The Fukuyama coupling is a coupling reaction taking place between a thioester and an organozinc halide in the presence of a palladium catalyst. The reaction product is a ketone. This reaction was disc… | Wikipedia: Category:Name reactions | |
| Glaser coupling Cross-coupling | The Glaser coupling is a type of coupling reaction. It is by far one of the oldest coupling reactions and is based on copper compounds like copper(I) chloride or copper(I) bromide and an additional ox… | Wikipedia: Category:Name reactions | |
| Heck–Matsuda reaction Cross-coupling | The Heck–Matsuda (HM) reaction is an organic reaction and a type of palladium catalysed arylation of olefins that uses arenediazonium salts as an alternative to aryl halides and triflates. | Wikipedia: Category:Name reactions | |
| Heck reaction Cross-coupling | The Heck reaction (also called the Mizoroki–Heck reaction) is the chemical reaction of an unsaturated halide (or triflate) with an alkene in the presence of a base and a palladium catalyst to form a … | Wikipedia: Category:Name reactions | |
| HECK REACTIONS OF ARYL CHLORIDES CATALYZED BY PALLADIUM/TRI-tert-BUTYLPHOSPHINE: (E)-2-METHYL-3-PHENYLACRYLIC ACID BUTYL ESTER AND (E)-4-(2-PHENYLETHENYL)BENZONITRILE (2-Propenoic acid, 2-methyl-3-phenyl-, butyl ester and Benzonitrile, 4-[(1E)-2-phenylethenyl]) Cross-coupling | Preparation of 2-Propenoic acid, 2-methyl-3-phenyl-, butyl ester and Benzonitrile, 4-[(1E)-2-phenylethenyl]. Submitted by Adam F. Littke and Gregory C. Fu1. DOI: 10.15227/orgsyn.081.0063. | Organic Syntheses | |
| Hirao coupling Cross-coupling | In organic chemistry, Hirao coupling is a chemical reaction for the formation of carbon-phosphorus bonds using palladium cross-coupling. Hirao coupling expands the scope of carbon-phosphorus bond form… | Wikipedia: Category:Name reactions | |
| Hiyama coupling Cross-coupling | The Hiyama coupling is a palladium-catalyzed cross-coupling reaction of organosilanes with organic halides used in organic chemistry to form carbon–carbon bonds (C-C bonds). This reaction was discover… | Wikipedia: Category:Name reactions | |
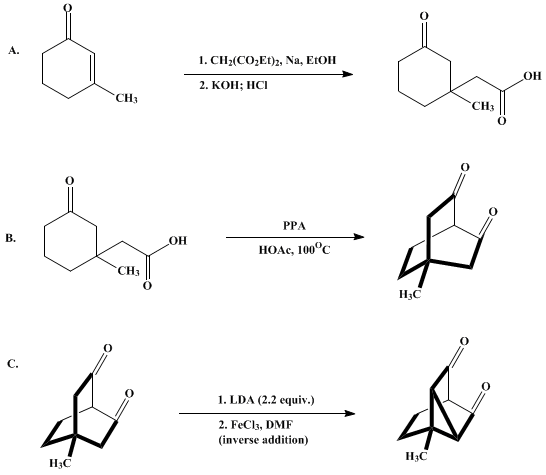
| INTRAMOLECULAR OXIDATIVE COUPLING OF A BISENOLATE: 4-METHYLTRICYCLO[2.2.2.03,5]OCTANE-2,6-DIONE (Tricyclo[3.2.1.02,7]octane-6,8-dione, 2-methyl-) Cross-coupling | Preparation of Tricyclo[3.2.1.02,7]octane-6,8-dione, 2-methyl-. Submitted by Marc-André Poupart, Gilbert Lassalle, and Leo A. Paquette1. DOI: 10.15227/orgsyn.069.0173. | Organic Syntheses | |
| Kumada coupling Cross-coupling | In organic chemistry, the Kumada coupling is a type of cross coupling reaction, useful for generating carbon–carbon bonds by the reaction of a Grignard reagent and an organic halide. The procedure use… | Wikipedia: Category:Name reactions | |
| Liebeskind–Srogl coupling Cross-coupling | The Liebeskind–Srogl coupling reaction is an organic reaction forming a new carbon–carbon bond from a thioester and a boronic acid using a metal catalyst. It is a cross-coupling reaction. This reactio… | Wikipedia: Category:Name reactions | |
| Murahashi coupling Cross-coupling | The Murahashi Coupling is a cross coupling reaction. The coupling partners are organolithiums and organic halides. Transition metal catalysts are required. The reaction was first reported by Shun-Ich… | Wikipedia: Category:Name reactions | |
| Negishi coupling Cross-coupling | The Negishi coupling is a widely employed transition metal catalyzed cross-coupling reaction. The reaction couples organic halides or triflates with organozinc compounds, forming carbon–carbon bonds … | Wikipedia: Category:Name reactions | |
| NICKEL-CATALYZED ASYMMETRIC NEGISHI CROSS-COUPLINGS OF RACEMIC SECONDARY ALLYLIC CHLORIDES WITH ALKYLZINCS: (S,E)-ETHYL 6-(1,3-DIOXOLAN-2-YL)-4-METHYLHEX-2-ENOATE (2-Hexenoic acid, 6-(1,3-dioxolan-2-yl)-4-methyl-, ethyl ester, (2E,4S)-) Cross-coupling | Preparation of 2-Hexenoic acid, 6-(1,3-dioxolan-2-yl)-4-methyl-, ethyl ester, (2E,4S)-. Submitted by Sha Lou and Gregory C. Fu1. DOI: 10.15227/orgsyn.087.0317. | Organic Syntheses | |
| Nickel-Catalyzed Cross-Coupling of Aryl Halides with Alkyl Halides: Ethyl 4-(4-(4-methylphenylsulfonamido)-phenyl)butanoate Cross-coupling | Preparation of Nickel-Catalyzed Cross-Coupling of Aryl Halides with Alkyl Halides: Ethyl 4-(4-(4-methylphenylsulfonamido)-phenyl)butanoate. Submitted by Daniel A.Everson, David T.George, and Daniel J.… | Organic Syntheses | |
| NICKEL-CATALYZED ENANTIOSELECTIVE NEGISHI CROSS-COUPLINGS OF RACEMIC SECONDARY α-BROMO AMIDES WITH ALKYLZINC REAGENTS: (S)-N-BENZYL-7-CYANO-2-ETHYL-N-PHENYLHEPTANAMIDE (Heptanamide, 7-cyano-2-ethyl-N-phenyl-N-(phenylmethyl)-, (2S)-) Cross-coupling | Preparation of Heptanamide, 7-cyano-2-ethyl-N-phenyl-N-(phenylmethyl)-, (2S)-. Submitted by Sha Lou and Gregory C. Fu1. DOI: 10.15227/orgsyn.087.0330. | Organic Syntheses | |
| ONE-POT DIAZOTIZATION AND HECK REACTION OF METHYL ANTHRANILATE: 2-(3-OXOPROPYL)BENZOIC ACID METHYL ESTER Cross-coupling | Preparation of ONE-POT DIAZOTIZATION AND HECK REACTION OF METHYL ANTHRANILATE: 2-(3-OXOPROPYL)BENZOIC ACID METHYL ESTER. Submitted by Florencio Zaragoza1. DOI: 10.15227/orgsyn.087.0226. | Organic Syntheses | |
| PALLADIUM-CATALYZED ALKYL-ALKYL SUZUKI CROSS-COUPLINGS OF PRIMARY ALKYL BROMIDES AT ROOM TEMPERATURE: (13-CHLOROTRIDECYLOXY)TRIETHYLSILANE (Silane, [(13-chlorotridecyl)oxy]triethyl-) Cross-coupling | Preparation of Silane, [(13-chlorotridecyl)oxy]triethyl-. Submitted by Sha Lou and Gregory C. Fu1. DOI: 10.15227/orgsyn.087.0299. | Organic Syntheses | |
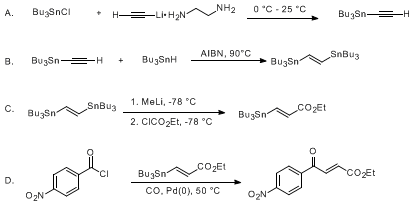
| PALLADIUM-CATALYZED COUPLING OF ACID CHLORIDES WITH ORGANOTIN REAGENTS: ETHYL (E)-4-(4-NITROPHENYL)-4-OXO-2-BUTENOATE (2-Butenoic acid, 4-(4-nitrophenyl)-4-oxo-, ethyl ester, (E)-) Cross-coupling | Preparation of 2-Butenoic acid, 4-(4-nitrophenyl)-4-oxo-, ethyl ester, (E)-. Submitted by A. F. Renaldo1, J. W. Labadie, and J. K. Stille2. DOI: 10.15227/orgsyn.067.0086. | Organic Syntheses | |
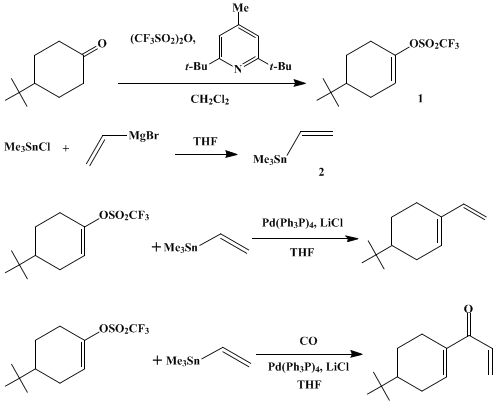
| PALLADIUM-CATALYZED COUPLING OF VINYL TRIFLATES WITH ORGANOSTANNANES: 4-tert-BUTYL-1-VINYLCYCLOHEXENE AND 1-(4-tert-BUTYLCYCLOHEXEN-1-YL)-2-PROPEN-1-ONE (Cyclohexene, 4-(1,1-dimethylethyl)-1-ethenyl- and 2-propen-1-one, 1-[4-(1,1-dimethylethyl)-1-cyclohexen-1-yl]) Cross-coupling | Preparation of Cyclohexene, 4-(1,1-dimethylethyl)-1-ethenyl- and 2-propen-1-one, 1-[4-(1,1-dimethylethyl)-1-cyclohexen-1-yl]. Submitted by William J. Scott1, G. T. Crisp2, and J. K. Stille3. DOI: 10.1… | Organic Syntheses | |
| PALLADIUM CATALYZED CROSS-COUPLING OF (Z)-1-HEPTENYLDIMETHYLSILANOL WITH 4-IODOANISOLE: (Z)-1-HEPTENYL)-4-METHOXYBENZENE (Benzene,1-(1Z)-1-heptenyl-4-methoxy-) Cross-coupling | Preparation of Benzene,1-(1Z)-1-heptenyl-4-methoxy-. Submitted by Scott E. Denmark and Zhigang Wang1. DOI: 10.15227/orgsyn.081.0042. | Organic Syntheses | |
| PALLADIUM-CATALYZED CROSS-COUPLING USING AN AIR-STABLE TRIMETHYLALUMINUM SOURCE. PREPARATION OF ETHYL 4-METHYLBENZOATE Cross-coupling | Preparation of PALLADIUM-CATALYZED CROSS-COUPLING USING AN AIR-STABLE TRIMETHYLALUMINUM SOURCE. PREPARATION OF ETHYL 4-METHYLBENZOATE. Submitted by Andrej Vinogradov and Simon Woodward1. DOI: 10.15227… | Organic Syntheses | |
| PLATINUM CATALYZED HYDROSILYLATION AND PALLADIUM CATALYZED CROSS-COUPLING: ONE-POT HYDROARYLATION OF 1-HEPTYNE TO (E)-1-(1-HEPTENYL)-4-METHOXYBENZENE (Benzene, 1-(1E)-1-heptenyl-4-methoxy-) Cross-coupling | Preparation of Benzene, 1-(1E)-1-heptenyl-4-methoxy-. Submitted by Scott E. Denmark and Zhigang Wang1. DOI: 10.15227/orgsyn.081.0054. | Organic Syntheses | |
| Preparation of 1-Monoacylglycerols via the Suzuki-Miyaura Reaction: 2,3-Dihydroxypropyl (Z)-tetradec-7-enoate Cross-coupling | Preparation of Preparation of 1-Monoacylglycerols via the Suzuki-Miyaura Reaction: 2,3-Dihydroxypropyl (Z)-tetradec-7-enoate. Submitted by Dexi Yang1, Valerie A. Cwynar1, David J. Hart1, Jakkam Madanm… | Organic Syntheses | |
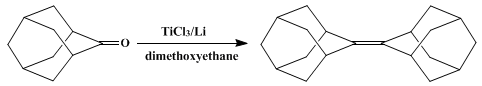
| REDUCTIVE COUPLING OF CARBONYLS TO ALKENES: ADAMANTYLIDENEADAMANTANE (Tricyclo[3.3.1.13,7]decane, tricyclo[3.3.1.13,7]decylidene-) Cross-coupling | Preparation of Tricyclo[3.3.1.13,7]decane, tricyclo[3.3.1.13,7]decylidene-. Submitted by Michael P. Fleming and John E. McMurry1. DOI: 10.15227/orgsyn.060.0113. | Organic Syntheses | |
| (S)-(+)-NEOMENTHYLDIPHENYLPHOSPHINE IN NICKEL-CATALYZED ASYMMETRIC REDUCTIVE COUPLING OF ALKYNES AND ALDEHYDES: ENANTIOSELECTIVE SYNTHESIS OF ALLYLIC ALCOHOLS AND α-HYDROXY KETONES ((R)-Butan-2-one, 1-cyclohexyl, 1-hydroxy-) Cross-coupling | Preparation of (R)-Butan-2-one, 1-cyclohexyl, 1-hydroxy-. Submitted by Aaron R. Van Dyke, Karen M. Miller, and Timothy F. Jamison1. DOI: 10.15227/orgsyn.084.0111. | Organic Syntheses | |
| Sonogashira coupling Cross-coupling | The Sonogashira reaction is a cross-coupling reaction used in organic synthesis to form carbon–carbon bonds. It employs a palladium catalyst as well as copper co-catalyst to form a carbon–carbon bond … | Wikipedia: Category:Name reactions | |
| STILLE COUPLINGS CATALYZED BY PALLADIUM-ON-CARBON WITH CuI AS A COCATALYST: SYNTHESIS OF 2-(4'-ACETYLPHENYL)THIOPHENE 1 Cross-coupling | Preparation of STILLE COUPLINGS CATALYZED BY PALLADIUM-ON-CARBON WITH CuI AS A COCATALYST: SYNTHESIS OF 2-(4'-ACETYLPHENYL)THIOPHENE 1. Submitted by Lanny S. Liebeskind2 and Eduardo Peña-Cabrera3. DOI… | Organic Syntheses | |
| Stille reaction Cross-coupling | The Stille reaction is a chemical reaction widely used in organic synthesis. The reaction involves the coupling of two organic groups, one of which is carried as an organotin compound (also known as o… | Wikipedia: Category:Name reactions | |
| SULFIDE CONTRACTION via ALKYLATIVE COUPLING: 3-METHYL-2,4-HEPTANEDIONE (2,4-Heptanedione, 3-methyl-) Cross-coupling | Preparation of 2,4-Heptanedione, 3-methyl-. Submitted by P. Loeliger1 and E. Flückiger2. DOI: 10.15227/orgsyn.055.0127. | Organic Syntheses | |
| Suzuki–Miyaura coupling: 4-bromobenzonitrile + phenylboronic acid → 4-cyanobiphenyl Scale Cross-coupling | Canonical Pd(0)-catalysed cross-coupling of an aryl halide with an aryl boronic acid. The 4-cyanobiphenyl target is the textbook demonstration substrate — electron-poor aryl halide + electron-rich bor… | Suzuki 1979 | |
| SUZUKI-MIYAURA CROSS-COUPLING: PREPARATION OF 2'-VINYLACETANILIDE (N-(2-Ethenylphenyl)acetamide) Cross-coupling | Preparation of N-(2-Ethenylphenyl)acetamide. Submitted by Bertrand Cottineau, Albane Kessler, and Donal F. O'Shea1. DOI: 10.15227/orgsyn.083.0045. | Organic Syntheses | |
| Suzuki reaction Cross-coupling | The Suzuki reaction or Suzuki coupling is an organic reaction that uses a palladium complex catalyst to cross-couple a boronic acid to an organohalide. It was first published in 1979 by Akira Suzuki, … | Wikipedia: Category:Name reactions | |
| Synthesis of 1,3-Dimethyl-3-(p-tolyl)-1H-pyrrolo[3,2-c]pyridin-2(3H)-one by Cu(II)-Mediated Direct Oxidative Coupling Cross-coupling | Preparation of Synthesis of 1,3-Dimethyl-3-(p-tolyl)-1H-pyrrolo[3,2-c]pyridin-2(3H)-one by Cu(II)-Mediated Direct Oxidative Coupling. Submitted by Chandan Dey and E. Peter Kündig*1. DOI: 10.15227/orgs… | Organic Syntheses | |
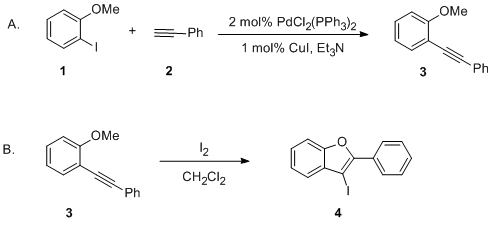
| Synthesis of 2,3-Disubstituted Benzofurans by the Palladium-Catalyzed Coupling of 2-Iodoanisoles and Terminal Alkynes, Followed by Electrophilic Cyclization: 3-Iodo-2-phenylbenzofuran Cross-coupling | Preparation of Synthesis of 2,3-Disubstituted Benzofurans by the Palladium-Catalyzed Coupling of 2-Iodoanisoles and Terminal Alkynes, Followed by Electrophilic Cyclization: 3-Iodo-2-phenylbenzofuran. … | Organic Syntheses | |
| SYNTHESIS OF (2R,3R)-2,3-DIMETHYL-1,4-BUTANEDIOL BY OXIDATIVE HOMOCOUPLING OF (4S)-ISOPROPYL-3-PROPIONYL-2-OXAZOLIDINONE (2,3-dimethylbutane-1,4-diol, (2R, 3R)-) Cross-coupling | Preparation of 2,3-dimethylbutane-1,4-diol, (2R, 3R)-. Submitted by Chong-Dao Lu and Armen Zakarian1. DOI: 10.15227/orgsyn.085.0158. | Organic Syntheses | |
| SYNTHESIS OF 2-SUBSTITUTED BIARYLS VIA Cu/Pd-CATALYZED DECARBOXYLATIVE CROSS-COUPLING OF 2-SUBSTITUTED POTASSIUM BENZOATES: 4-METHYL-2'-NITROBIPHENYL AND 2-ACETYL-4'-METHYLBIPHENYL (Biphenyl, 4-Methyl-2'-nitro-) Cross-coupling | Preparation of Biphenyl, 4-Methyl-2'-nitro-. Submitted by Lukas J. Gooßen1, Nuria Rodríguez, Christophe Linder, Bettina Zimmermann, and Thomas Knauber. DOI: 10.15227/orgsyn.085.0196. | Organic Syntheses | |
| SYNTHESIS OF 3-PYRIDYLBORONIC ACID AND ITS PINACOL ESTER. APPLICATION OF 3-PYRIDYLBORONIC ACID IN SUZUKI COUPLING TO PREPARE 3-PYRIDIN-3-YLQUINOLINE (Quinoline, 3-(3-pyridinyl)-) Cross-coupling | Preparation of Quinoline, 3-(3-pyridinyl)-. Submitted by Wenjie Li, Dorian P. Nelson, Mark S. Jensen, R. Scott Hoerrner, Dongwei Cai, and Robert D. Larsen1. DOI: 10.15227/orgsyn.081.0089. | Organic Syntheses |