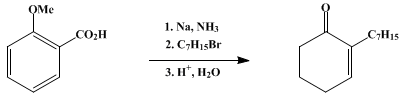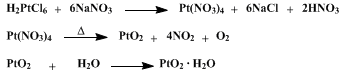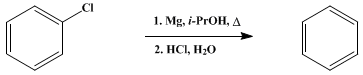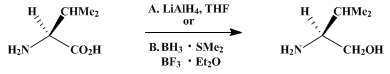Browse · Reactions
Reactions
58 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
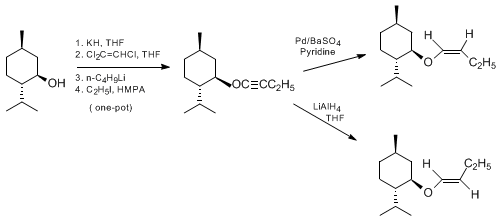
| ACETYLENIC ETHERS FROM ALCOHOLS AND THEIR REDUCTION TO Z- AND E-ENOL ETHERS: PREPARATION OF 1-MENTHOXY-1-BUTYNE FROM MENTHOL AND CONVERSION TO (Z)- AND (E)-1-MENTHOXY-1-BUTENE ([Cyclohexane, 2-(1-butynyloxy)-4-methyl-1-(1-methylethyl)-[1S-(1α,2β,4β)]-], and [[[Cyclohexane, 2-(1-butenyloxy)-4-methyl-1-(1-methylethyl)-, [1S-[1α,2β(Z),4β]]- and [1S-[1α,2β(E),4β]]-) Reduction | Preparation of [Cyclohexane, 2-(1-butynyloxy)-4-methyl-1-(1-methylethyl)-[1S-(1α,2β,4β)]-], and [[[Cyclohexane, 2-(1-butenyloxy)-4-methyl-1-(1-methylethyl)-, [1S-[1α,2β(Z),4β]]- and [1S-[1α,2β(E),4β]]… | Organic Syntheses | |
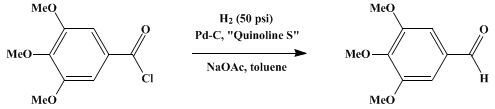
| ALDEHYDES FROM ACID CHLORIDES BY MODIFIED ROSENMUND REDUCTION: 3,4,5-TRIMETHOXYBENZALDEHYDE (Benzaldehyde, 3,4,5-trimethoxy-) Reduction | Preparation of Benzaldehyde, 3,4,5-trimethoxy-. Submitted by A. I. Rachlin, H. Gurien, and D. P. Wagner1. DOI: 10.15227/orgsyn.051.0008. | Organic Syntheses | |
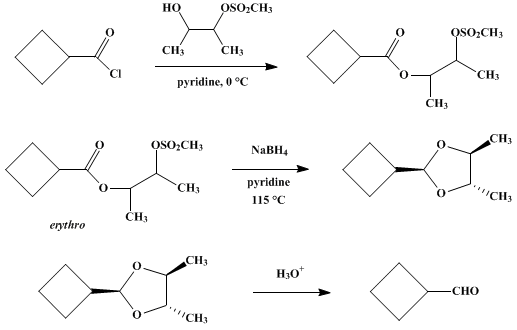
| ALDEHYDES FROM ACID CHLORIDES BY REDUCTION OF ESTER-MESYLATES WITH SODIUM BOROHYDRIDE: CYCLOBUTANECARBOXALDEHYDE Reduction | Preparation of ALDEHYDES FROM ACID CHLORIDES BY REDUCTION OF ESTER-MESYLATES WITH SODIUM BOROHYDRIDE: CYCLOBUTANECARBOXALDEHYDE. Submitted by M. Ross Johnson and Bruce Rickborn1. DOI: 10.15227/orgsyn.… | Organic Syntheses | |
| ALKYLATION OF THE ANION FROM BIRCH REDUCTION OF o-ANISIC ACID: 2-HEPTYL-2-CYCLOHEXENONE Reduction | Preparation of ALKYLATION OF THE ANION FROM BIRCH REDUCTION OF o-ANISIC ACID: 2-HEPTYL-2-CYCLOHEXENONE. Submitted by D. F. Taber, B. P. Gunn, and I-Ching Chiu1. DOI: 10.15227/orgsyn.061.0059. | Organic Syntheses | |
| APPARATUS FOR CATALYTIC REDUCTION Reduction | Preparation of APPARATUS FOR CATALYTIC REDUCTION. Submitted by Roger Adams and V. Voorhees. DOI: 10.15227/orgsyn.008.0010. | Organic Syntheses | |
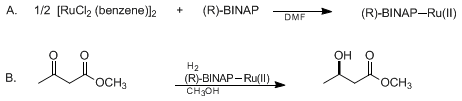
| ASYMMETRIC HYDROGENATION OF 3-OXO CARBOXYLATES USING BINAP-RUTHENIUM COMPLEXES: (R)-(−)-METHYL 3-HYDROXYBUTANOATE (Butanoic acid, 3-hydroxy-, methyl ester, (R)-) Reduction | Preparation of Butanoic acid, 3-hydroxy-, methyl ester, (R)-. Submitted by Masato Kitamura, Makoto Tokunaga, Takeshi Ohkuma, and Ryoji Noyori1. DOI: 10.15227/orgsyn.071.0001. | Organic Syntheses | |
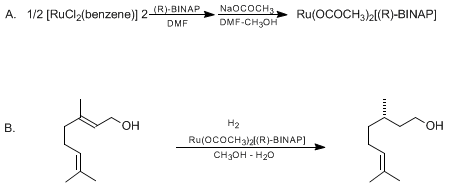
| ASYMMETRIC HYDROGENATION OF ALLYLIC ALCOHOLS USING BINAP-RUTHENIUM COMPLEXES: (S)-(−)-CITRONELLOL (6-Octen-1-ol, 3,7-dimethyl, (S)-) Reduction | Preparation of 6-Octen-1-ol, 3,7-dimethyl, (S)-. Submitted by Hidemasa Takaya1, Tetsuo Ohta1, Shin-ichi Inoue2, Makoto Tokunaga2, Masato Kitamura2, and Ryoji Noyori2. DOI: 10.15227/orgsyn.072.0074. | Organic Syntheses | |
| ASYMMETRIC HYDROGENATION OF KETOPANTOYL LACTONE: D-(−)-PANTOYL LACTONE (2(3H)-Furanone, dihydro-3-hydroxy-4,4-dimethyl-) Reduction | Preparation of 2(3H)-Furanone, dihydro-3-hydroxy-4,4-dimethyl-. Submitted by I. Ojima, T. Kogure, and Y. Yoda1. DOI: 10.15227/orgsyn.063.0018. | Organic Syntheses | |
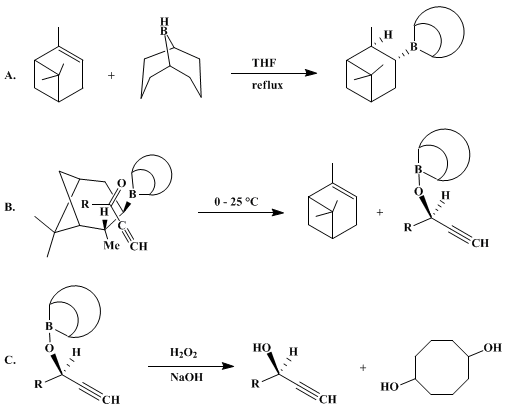
| ASYMMETRIC REDUCTION OF α,β-ACETYLENIC KETONES WITH B-3-PINANYL-9-BORABICYCLO[3.3.1]NONANE: (R)-( + )-1-OCTYN-3-OL (1-Octyn-3-ol, (R)-) Reduction | Preparation of 1-Octyn-3-ol, (R)-. Submitted by M. Mark Midland and Richard S. Graham1. DOI: 10.15227/orgsyn.063.0057. | Organic Syntheses | |
| Béchamp reduction Reduction | The Béchamp reduction (or Béchamp process) is a chemical reaction that converts aromatic nitro compounds to their corresponding anilines using iron as the reductant: | Wikipedia: Category:Name reactions | |
| Birch reduction Reduction | The Birch reduction or Metal-Ammonia reduction is an organic reaction that is used to convert arenes to 1,4-cyclohexadienes. The reaction is named after the Australian chemist Arthur Birch and involve… | Wikipedia: Category:Name reactions | |
| Bouveault–Blanc reduction Reduction | The Bouveault–Blanc reduction is a chemical reaction in which an ester is reduced to primary alcohols using absolute ethanol and sodium metal. It was first reported by Louis Bouveault and Gustave Lou… | Wikipedia: Category:Name reactions | |
| CATALYTIC ENANTIOSELECTIVE BORANE REDUCTION OF BENZYL OXIMES: PREPARATION OF (S)-1-PYRIDIN-3-YL-ETHYLAMINE BIS HYDROCHLORIDE Reduction | Preparation of CATALYTIC ENANTIOSELECTIVE BORANE REDUCTION OF BENZYL OXIMES: PREPARATION OF (S)-1-PYRIDIN-3-YL-ETHYLAMINE BIS HYDROCHLORIDE. Submitted by Kun Huang and Margarita Ortiz-Marciales1. DOI:… | Organic Syntheses | |
| CATALYTIC REDUCTION OF AMIDES TO AMINES WITH HYDROSILANES USING A TRIRUTHENIUM CARBONYL CLUSTER AS THE CATALYST Reduction | Preparation of CATALYTIC REDUCTION OF AMIDES TO AMINES WITH HYDROSILANES USING A TRIRUTHENIUM CARBONYL CLUSTER AS THE CATALYST. Submitted by Yukihiro Motoyama, Chikara Itonaga, Toshiki Ishida, Mikihir… | Organic Syntheses | |
| Clemmensen reduction Reduction | Clemmensen reduction is a chemical reaction described as a reduction of ketones or aldehydes to alkanes using zinc amalgam and concentrated hydrochloric acid (HCl). This reaction is named after Erik C… | Wikipedia: Category:Name reactions | |
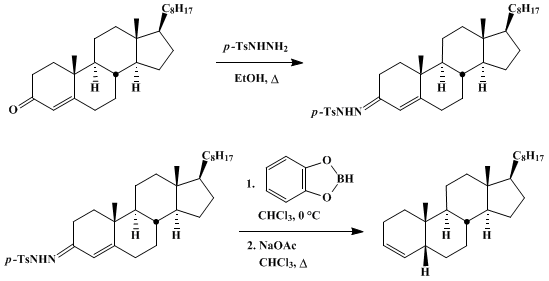
| CONJUGATE REDUCTION OF α,β-UNSATURATED p-TOLUENESULFONYLHYDRAZONES TO ALKENES WITH CATECHOLBORANE: 5β-CHOLEST-3-ENE (Cholest-3-ene, (5β)-) Reduction | Preparation of Cholest-3-ene, (5β)-. Submitted by George W. Kabalka1, Robert Hutchins2, Nicholas R. Natale2, Dominic T. C. Yang3, and Vicky Broach3. DOI: 10.15227/orgsyn.059.0042. | Organic Syntheses | |
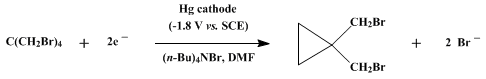
| CONTROLLED-POTENTIAL ELECTROLYTIC REDUCTION: 1,1-BIS(BROMOMETHYL)CYCLOPROPANE (Cyclopropane, 1,1-bis(bromomethyl)-) Reduction | Preparation of Cyclopropane, 1,1-bis(bromomethyl)-. Submitted by M. R. Rifi1. DOI: 10.15227/orgsyn.052.0022. | Organic Syntheses | |
| Corey–Itsuno reduction Reduction | The Corey–Itsuno reduction, also known as the Corey–Bakshi–Shibata (CBS) reduction, is a chemical reaction in which a prochiral ketone is enantioselectively reduced to produce the corresponding chiral… | Wikipedia: Category:Name reactions | |
| DIRECTED HOMOGENEOUS HYDROGENATION: METHYL anti-3-HYDROXY-2-METHYLPENTANOATE (Pentanoic acid, 3-hydroxy-2-methyl-, methyl ester, (R*, R*)-(±)-) Reduction | Preparation of Pentanoic acid, 3-hydroxy-2-methyl-, methyl ester, (R*, R*)-(±)-. Submitted by John M. Brown, Phillip L. Evans, and Alun P. James1. DOI: 10.15227/orgsyn.068.0064. | Organic Syntheses | |
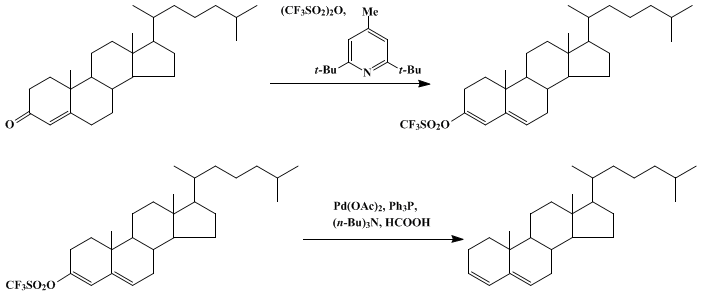
| Discussion Addendum for: PALLADIUM-CATALYZED REDUCTION OF VINYL TRIFLUOROMETHANESULFONATES TO ALKENES: CHOLESTA-3,5-DIENE Reduction | Preparation of Discussion Addendum for: PALLADIUM-CATALYZED REDUCTION OF VINYL TRIFLUOROMETHANESULFONATES TO ALKENES: CHOLESTA-3,5-DIENE. Submitted by Sandro Cacchi, Enrico Morera, and Giorgio Ortar.1… | Organic Syntheses | |
| ENONE REDUCTION-ENOLATE ALKYLATION SEQUENCE: 2-ALLYL-3-METHYLCYCLOHEXANONE (Cyclohexanone, 3-methyl-2-(3-propenyl)-) Reduction | Preparation of Cyclohexanone, 3-methyl-2-(3-propenyl)-. Submitted by Drury Caine1, Sam T. Chao1, and Homer A. Smith2. DOI: 10.15227/orgsyn.056.0052. | Organic Syntheses | |
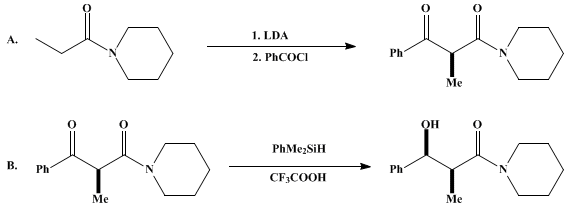
| erythro-DIRECTED REDUCTION OF A β-KETO AMIDE: ERYTHRO-1-(3-HYDROXY-2-METHYL-3-PHENYLPROPANOYL)PIPERIDINE (Piperidine, 1-(3-hydroxy-2-methyl-1-oxo-3-phenylpropyl)-, (R*, R*)-(±)-) Reduction | Preparation of Piperidine, 1-(3-hydroxy-2-methyl-1-oxo-3-phenylpropyl)-, (R*, R*)-(±)-. Submitted by M. Fujita and T. Hiyama1. DOI: 10.15227/orgsyn.069.0044. | Organic Syntheses | |
| Evans–Saksena reduction Reduction | The Saksena–Evans reduction is a diastereoselective reduction of β-hydroxy ketones to the corresponding anti-dialcohols, employing the reagent tetramethylammonium triacetoxyborohydride (Me4NHB(OAc)3).… | Wikipedia: Category:Name reactions | |
| Fukuyama reduction Reduction | The Fukuyama reduction is an organic reaction and an organic reduction in which a thioester is reduced to an aldehyde by a silyl hydride in presence of a catalytic amount of palladium. This reaction w… | Wikipedia: Category:Name reactions | |
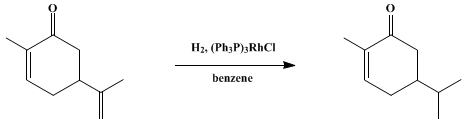
| HOMOGENEOUS CATALYTIC HYDROGENATION: DIHYDROCARVONE (2-Cyclohexen-1-one, 2-methyl-5-(1-methylethyl)-) Reduction | Preparation of 2-Cyclohexen-1-one, 2-methyl-5-(1-methylethyl)-. Submitted by Robert E. Ireland1 and P. Bey2. DOI: 10.15227/orgsyn.053.0063. | Organic Syntheses | |
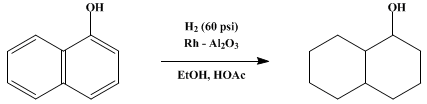
| HYDROGENATION OF AROMATIC NUCLEI: 1-DECALOL Reduction | Preparation of HYDROGENATION OF AROMATIC NUCLEI: 1-DECALOL. Submitted by A. I. Meyers1, W. N. Beverung, and R. Gault2. DOI: 10.15227/orgsyn.051.0103. | Organic Syntheses | |
| INDIUM/AMMONIUM CHLORIDE-MEDIATED SELECTIVE REDUCTION OF AROMATIC NITRO COMPOUNDS: ETHYL 4-AMINOBENZOATE ((Benzoic acid, 4-amino-, ethyl ester)) Reduction | Preparation of (Benzoic acid, 4-amino-, ethyl ester). Submitted by Bimal K. Banik1, Indrani Banik, and Frederick F. Becker. DOI: 10.15227/orgsyn.081.0188. | Organic Syntheses | |
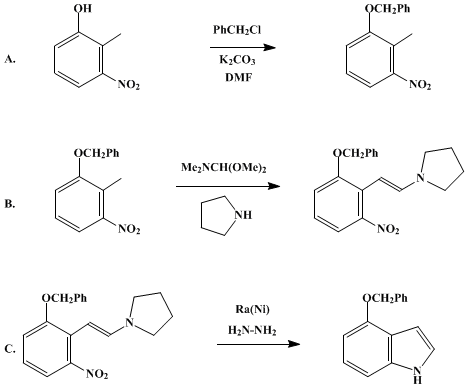
| INDOLES FROM 2-METHYLNITROBENZENES BY CONDENSATION WITH FORMAMIDE ACETALS FOLLOWED BY REDUCTION: 4-BENZYLOXYINDOLE (1H-Indole, 4-(phenylmethoxy)-) Reduction | Preparation of 1H-Indole, 4-(phenylmethoxy)-. Submitted by Andrew D. Batcho1 and Willy Leimgruber2. DOI: 10.15227/orgsyn.063.0214. | Organic Syntheses | |
| Luche reduction Reduction | Luche reduction is the selective organic reduction of α,β-unsaturated ketones to allylic alcohols. The active reductant is described as "cerium borohydride", which is generated in situ from NaBH4 and … | Wikipedia: Category:Name reactions | |
| Meerwein–Ponndorf–Verley reduction Reduction | The Meerwein–Ponndorf–Verley (MPV) reduction in organic chemistry is the reduction of ketones and aldehydes to their corresponding alcohols utilizing aluminium alkoxide catalysis in the presence of a … | Wikipedia: Category:Name reactions | |
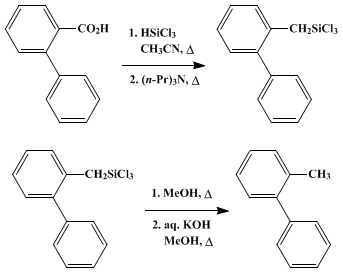
| METHYL GROUPS BY REDUCTION OF AROMATIC CARBOXYLIC ACIDS WITH TRICHLOROSILANE - TRI-n-PROPYLAMINE: 2-METHYLBIPHENYL (1,1'-Biphenyl, 2-methyl) Reduction | Preparation of 1,1'-Biphenyl, 2-methyl. Submitted by George S. Li, David F. Ehler, and R. A. Benkeser1. DOI: 10.15227/orgsyn.056.0083. | Organic Syntheses | |
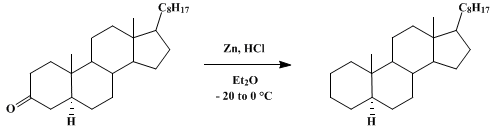
| MODIFIED CLEMMENSEN REDUCTION: CHOLESTANE Reduction | Preparation of MODIFIED CLEMMENSEN REDUCTION: CHOLESTANE. Submitted by Shosuke Yamamura1, Masaaki Toda2, and Yoshimasa Hirata2. DOI: 10.15227/orgsyn.053.0086. | Organic Syntheses | |
| Mozingo reduction Reduction | The Mozingo reduction, also known as Mozingo reaction or thioketal reduction, is a chemical reaction capable of fully reducing a ketone or aldehyde to the corresponding alkane via a dithioacetal. The … | Wikipedia: Category:Name reactions | |
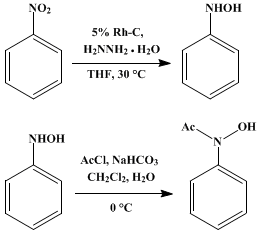
| N-ACETYL-N-PHENYLHYDROXYLAMINE VIA CATALYTIC TRANSFER HYDROGENATION OF NITROBENZENE USING HYDRAZINE AND RHODIUM ON CARBON (Acetamide, N-hydroxy-N-phenyl-) Reduction | Preparation of Acetamide, N-hydroxy-N-phenyl-. Submitted by P. W. Oxley, B. M. Adger, M. J. Sasse, and M. A. Forth1. DOI: 10.15227/orgsyn.067.0187. | Organic Syntheses | |
| Narasaka–Prasad reduction Reduction | The Narasaka–Prasad reduction, sometimes simply called Narasaka reduction, is a diastereoselective reduction of β-hydroxy ketones to the corresponding syn-dialcohols. The reaction employs a boron chel… | Wikipedia: Category:Name reactions | |
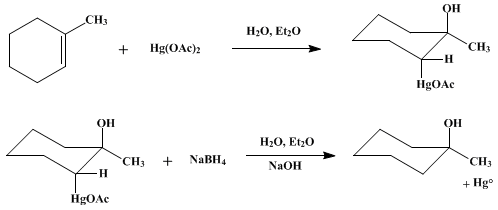
| OXYMERCURATION-REDUCTION: ALCOHOLS FROM OLEFINS: 1-METHYLCYCLOHEXANOL (Cyclohexanol, 1-methyl-) Reduction | Preparation of Cyclohexanol, 1-methyl-. Submitted by J. M. Jerkunica and T. G. Traylor1. DOI: 10.15227/orgsyn.053.0094. | Organic Syntheses | |
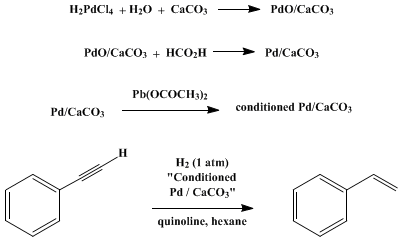
| PALLADIUM CATALYST FOR PARTIAL REDUCTION OF ACETYLENES1 Reduction | Preparation of PALLADIUM CATALYST FOR PARTIAL REDUCTION OF ACETYLENES1. Submitted by H. Lindlar and R. Dubuis2. DOI: 10.15227/orgsyn.046.0089. | Organic Syntheses | |
| PALLADIUM-CATALYZED REDUCTION OF VINYL TRIFLUOROMETHANESULFONATES TO ALKENES: CHOLESTA-3,5-DIENE Reduction | Preparation of PALLADIUM-CATALYZED REDUCTION OF VINYL TRIFLUOROMETHANESULFONATES TO ALKENES: CHOLESTA-3,5-DIENE. Submitted by Sandro Cacchi1, Enrico Morera2, and Giorgio Ortar2. DOI: 10.15227/orgsyn.0… | Organic Syntheses | |
| PLATINUM CATALYST FOR REDUCTIONS (Platinic oxide) Reduction | Preparation of Platinic oxide. Submitted by Roger Adams, V. Voorhees, and R. L. Shriner. DOI: 10.15227/orgsyn.008.0092. | Organic Syntheses | |
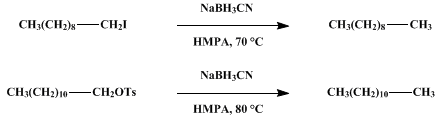
| REDUCTION OF ALKYL HALIDES AND TOSYLATES WITH SODIUM CYANOBOROHYDRIDE IN HEXAMETHYLPHOSPHORIC TRIAMIDE (HMPA):A. 1-IODODECANE TO n-DECANEB. 1-DODECYL TOSYLATE TO n-DODECANE Reduction | Preparation of REDUCTION OF ALKYL HALIDES AND TOSYLATES WITH SODIUM CYANOBOROHYDRIDE IN HEXAMETHYLPHOSPHORIC TRIAMIDE (HMPA):A. 1-IODODECANE TO n-DECANEB. 1-DODECYL TOSYLATE TO n-DODECANE. Submitted b… | Organic Syntheses | |
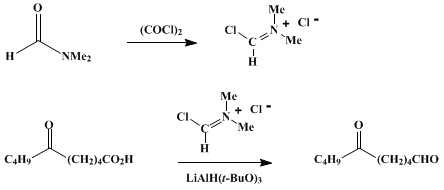
| REDUCTION OF CARBOXYLIC ACIDS TO ALDEHYDES: 6-OXODECANAL (Decanal, 6-oxo-) Reduction | Preparation of Decanal, 6-oxo-. Submitted by Tamotsu Fujisawa and Toshio Sato1. DOI: 10.15227/orgsyn.066.0121. | Organic Syntheses | |
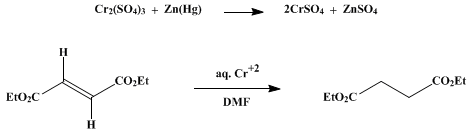
| REDUCTION OF CONJUGATED ALKENES WITH CHROMIUM(II) SULFATE: DIETHYL SUCCINATE (Succinic acid, diethyl ester) Reduction | Preparation of Succinic acid, diethyl ester. Submitted by A. Zurqiyah and C. E. Castro1. DOI: 10.15227/orgsyn.049.0098. | Organic Syntheses | |
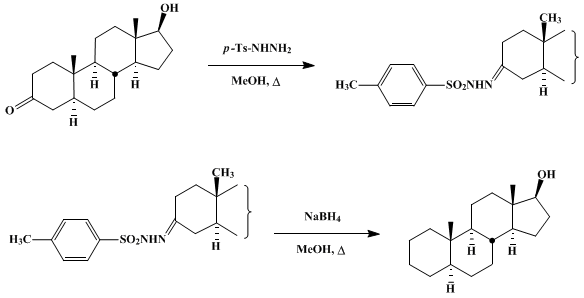
| REDUCTION OF KETONES BY USE OF THE TOSYLHYDRAZONE DERIVATIVES: ANDROSTAN-17 β-OL (Androstan-17-ol, (5α, 17β)-) Reduction | Preparation of Androstan-17-ol, (5α, 17β)-. Submitted by L. Caglioti1. DOI: 10.15227/orgsyn.052.0122. | Organic Syntheses | |
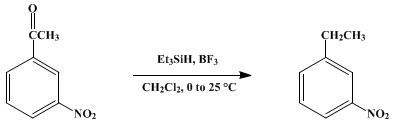
| REDUCTION OF KETONES TO HYDROCARBONS WITH TRIETHYLSILANE: m-NITROETHYLBENZENE (Benzene, 1-ethyl-3-nitro-) Reduction | Preparation of Benzene, 1-ethyl-3-nitro-. Submitted by James L. Fry, Steven B. Silverman, and Michael Orfanopoulos1. DOI: 10.15227/orgsyn.060.0108. | Organic Syntheses | |
| REDUCTION OF ORGANIC HALIDES. CHLOROBENZENE TO BENZENE Reduction | Preparation of REDUCTION OF ORGANIC HALIDES. CHLOROBENZENE TO BENZENE. Submitted by D. Bryce-Smith and B. J. Wakefield1. DOI: 10.15227/orgsyn.047.0103. | Organic Syntheses | |
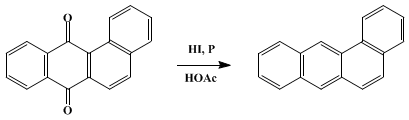
| REDUCTION OF QUINONES WITH HYDRIODIC ACID: BENZ[a]ANTHRACENE Reduction | Preparation of REDUCTION OF QUINONES WITH HYDRIODIC ACID: BENZ[a]ANTHRACENE. Submitted by Maria Konieczny and Ronald G. Harvey1. DOI: 10.15227/orgsyn.062.0165. | Organic Syntheses | |
| REDUCTION OF SULFONYL HALIDES WITH ZINC POWDER: S-METHYL METHANETHIOSULFONATE (Methanesulfonothioic acid, S-methyl ester) Reduction | Preparation of Methanesulfonothioic acid, S-methyl ester. Submitted by Fabrice Chemla1 and Philippe Karoyan2. DOI: 10.15227/orgsyn.078.0099. | Organic Syntheses | |
| REDUCTION OF α-AMINO ACIDS: L-VALINOL (1-Butanol, 2-amino-3-methyl-, (S)-) Reduction | Preparation of 1-Butanol, 2-amino-3-methyl-, (S)-. Submitted by D. A. Dickman1, A. I. Meyers1, G. A. Smith2, and R. E. Gawley2. DOI: 10.15227/orgsyn.063.0136. | Organic Syntheses | |
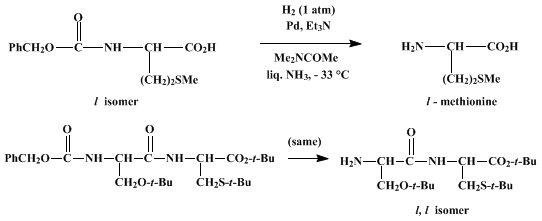
| REMOVAL OF Nα-BENZYLOXYCARBONYL GROUPS FROM SULFUR-CONTAINING PEPTIDES BY CATALYTIC HYDROGENATION IN LIQUID AMMONIA: O-tert-BUTYL-L-SERYL-S-tert-BUTYL-L-CYSTEINE tert-BUTYL ESTER Reduction | Preparation of REMOVAL OF Nα-BENZYLOXYCARBONYL GROUPS FROM SULFUR-CONTAINING PEPTIDES BY CATALYTIC HYDROGENATION IN LIQUID AMMONIA: O-tert-BUTYL-L-SERYL-S-tert-BUTYL-L-CYSTEINE tert-BUTYL ESTER. Submi… | Organic Syntheses | |
| Rosenmund reduction Reduction | The Rosenmund reduction is a hydrogenation process in which an acyl chloride is selectively reduced to an aldehyde. The reaction was named after Karl Wilhelm Rosenmund, who first reported it in 1918. | Wikipedia: Category:Name reactions |