Reactions · Organic Syntheses
ALDEHYDES FROM ACID CHLORIDES BY MODIFIED ROSENMUND REDUCTION: 3,4,5-TRIMETHOXYBENZALDEHYDE (Benzaldehyde, 3,4,5-trimethoxy-)
Reduction
Overview
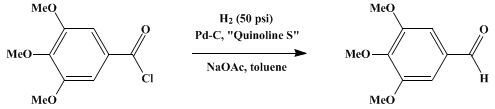
Preparation of Benzaldehyde, 3,4,5-trimethoxy-. Submitted by A. I. Rachlin, H. Gurien, and D. P. Wagner1. DOI: 10.15227/orgsyn.051.0008.
Actions
- This entry doesn't have stoichiometric equivalents on file yet — the Reaction Scale Calculator needs at least one participant tagged with an equivalents value.
- Mechanism / source ↗
- View as JSON ↗
Search literature
More Reduction reactions
- ACETYLENIC ETHERS FROM ALCOHOLS AND THEIR REDUCTION TO Z- AND E-ENOL ETHERS: PREPARATION OF 1-MENTHOXY-1-BUTYNE FROM MENTHOL AND CONVERSION TO (Z)- AND (E)-1-MENTHOXY-1-BUTENE ([Cyclohexane, 2-(1-butynyloxy)-4-methyl-1-(1-methylethyl)-[1S-(1α,2β,4β)]-], and [[[Cyclohexane, 2-(1-butenyloxy)-4-methyl-1-(1-methylethyl)-, [1S-[1α,2β(Z),4β]]- and [1S-[1α,2β(E),4β]]-) Reduction
- ALDEHYDES FROM ACID CHLORIDES BY REDUCTION OF ESTER-MESYLATES WITH SODIUM BOROHYDRIDE: CYCLOBUTANECARBOXALDEHYDE Reduction
- ALKYLATION OF THE ANION FROM BIRCH REDUCTION OF o-ANISIC ACID: 2-HEPTYL-2-CYCLOHEXENONE Reduction
- APPARATUS FOR CATALYTIC REDUCTION Reduction
- ASYMMETRIC HYDROGENATION OF 3-OXO CARBOXYLATES USING BINAP-RUTHENIUM COMPLEXES: (R)-(−)-METHYL 3-HYDROXYBUTANOATE (Butanoic acid, 3-hydroxy-, methyl ester, (R)-) Reduction
- ASYMMETRIC HYDROGENATION OF ALLYLIC ALCOHOLS USING BINAP-RUTHENIUM COMPLEXES: (S)-(−)-CITRONELLOL (6-Octen-1-ol, 3,7-dimethyl, (S)-) Reduction