Reactions · Organic Syntheses ↗
NUCLEOPHILIC HYDROXYMETHYLATION BY THE (ISOPROPOXYDIMETHYLSILYL)METHYL GRIGNARD REAGENT: 1-(HYDROXYMETHYL)CYCLOHEXANOL FROM CYCLOHEXANONE (Cyclohexanemethanol, 1-hydroxy-)
Organometallic addition
Overview
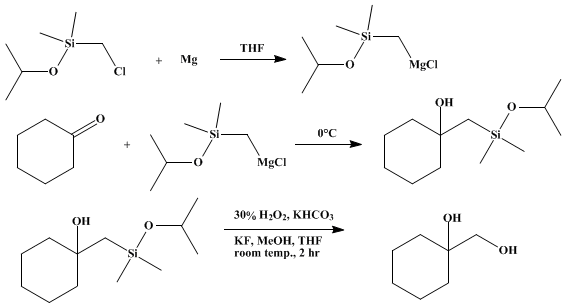
Preparation of Cyclohexanemethanol, 1-hydroxy-. Submitted by Kohei Tamao, Neyoshi Ishida, Yoshihiko Ito, and Makoto Kumada1. DOI: 10.15227/orgsyn.069.0096.
Actions
- This entry doesn't have stoichiometric equivalents on file yet — the Reaction Scale Calculator needs at least one participant tagged with an equivalents value.
- Mechanism / source ↗
- View as JSON ↗
Search literature
More Organometallic addition reactions
- ALDEHYDES FROM 4,4-DIMETHYL-2-OXAZOLINE AND GRIGNARD REAGENTS: o-ANISALDEHYDE (Benzaldehyde, 2-methoxy-) Organometallic addition
- Barbier reaction Organometallic addition
- Barbier–Wieland degradation Organometallic addition
- Cross-Coupling of Alkenyl/Aryl Carboxylates with Grignard Reagents via Fe-Catalyzed C-O Bond Activation Organometallic addition
- FORMYL TRANSFER TO GRIGNARD REAGENTS WITH N-FORMYLPIPERIDINE: 3-PHENYLPROPIONALDEHYDE Organometallic addition
- Grignard addition: phenylmagnesium bromide + benzaldehyde → diphenylmethanol Organometallic addition