Browse · Reactions
Reactions
15 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| 1,2-Wittig rearrangement Olefination | A 1,2-Wittig rearrangement is a categorization of chemical reactions in organic chemistry, and consists of a 1,2-rearrangement of an ether with an alkyllithium compound. The reaction is named for Nobe… | Wikipedia: Category:Name reactions | |
| 2,3-Wittig rearrangement Olefination | The [2,3]-Wittig rearrangement is the transformation of an allylic ether into a homoallylic alcohol via a concerted, pericyclic process. Because the reaction is concerted, it exhibits a high degree of… | Wikipedia: Category:Name reactions | |
| Aza-Wittig reaction Olefination | The Aza-Wittig reaction or is a chemical reaction of a carbonyl group with an aza-ylide, also known as an iminophosphorane (R3P=NR'). Aza-Wittig reactions are most commonly used to convert aldehydes… | Wikipedia: Category:Name reactions | |
| GENERATION OF YNOLATE AND Z-SELECTIVE OLEFINATION OF ACYLSILANES: (Z)-2-METHYL-3-TRIMETHYLSILYL-2-BUTENOIC ACID ((Z)-2-Butenoic acid, 2-methyl-3-trimethylsilyl-) Olefination | Preparation of (Z)-2-Butenoic acid, 2-methyl-3-trimethylsilyl-. Submitted by Mitsuru Shindo1, Kenji Matsumoto2, and Kozo Shishido2. DOI: 10.15227/orgsyn.084.0011. | Organic Syntheses | |
| Julia olefination Olefination | The Julia olefination (also known as the Julia–Lythgoe olefination) is the chemical reaction used in organic chemistry of phenyl sulfones (1) with aldehydes (or ketones) to give alkenes (olefins)(3) a… | Wikipedia: Category:Name reactions | |
| Kauffmann olefination Olefination | The Kauffmann olefination is a chemical reaction to convert aldehydes and ketones to olefins with a terminal methylene group. This reaction was discovered by the German chemist Thomas Kauffmann and is… | Wikipedia: Category:Name reactions | |
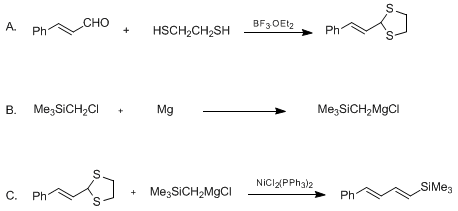
| NICKEL-CATALYZED SILYLOLEFINATION OF ALLYLIC DITHIOACETALS: (E,E)-TRIMETHYL(4-PHENYL-1,3-BUTADIENYL)SILANE (Silane, trimethyl (4-phenyl-1,3-butadienyl)-, (E,E)-) Olefination | Preparation of Silane, trimethyl (4-phenyl-1,3-butadienyl)-, (E,E)-. Submitted by Zhi-Jie Ni and Tien-Yau Luh1. DOI: 10.15227/orgsyn.070.0240. | Organic Syntheses | |
| Peterson olefination Olefination | The Peterson olefination (also called the Peterson reaction) is the chemical reaction of α-silyl carbanions (1 in diagram below) with ketones (or aldehydes) to form a β-hydroxysilane (2) which elimina… | Wikipedia: Category:Name reactions | |
| PREPARATION OF HORNER-WADSWORTH-EMMONS REAGENT: METHYL 2-BENZYLOXYCARBONYLAMINO-2- (DIMETHOXY- PHOSPHINYL)ACETATE Olefination | Preparation of PREPARATION OF HORNER-WADSWORTH-EMMONS REAGENT: METHYL 2-BENZYLOXYCARBONYLAMINO-2- (DIMETHOXY- PHOSPHINYL)ACETATE. Submitted by Hiroki Azuma1, Kentaro Okano1, Tohru Fukuyama2, and Hidet… | Organic Syntheses | |
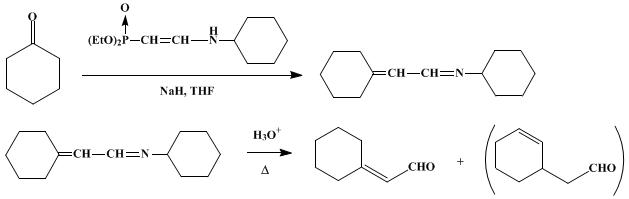
| PREPARATION OF α,β-UNSATURATED ALDEHYDES via THE WITTIG REACTION: CYCLOHEXYLIDENEACETALDEHYDE (Acetaldehyde, cyclohexylidene-) Olefination | Preparation of Acetaldehyde, cyclohexylidene-. Submitted by Wataru Nagata1, Toshio Wakabayashi2, and Yoshio Hayase1. DOI: 10.15227/orgsyn.053.0104. | Organic Syntheses | |
| SYNTHESIS OF (−)-(E,S)-3-(BENZYLOXY)-1-BUTENYL PHENYL SULFONE VIA A HORNER-WADSWORTH-EMMONS REACTION OF (−)-(S)-2-(BENZYLOXY)PROPANAL (Benzene, [[[1-methyl-3-(phenylsulfonyl)-2-propenyl]oxy]methyl]-, [S-(E)]-) from (Propanal, 2-(phenylmethoxy)-, (S)-)) Olefination | Preparation of Benzene, [[[1-methyl-3-(phenylsulfonyl)-2-propenyl]oxy]methyl]-, [S-(E)]-) from (Propanal, 2-(phenylmethoxy)-, (S)-). Submitted by D. Enders1 , S. von Berg1 , and B. Jandeleit2. DOI: 10… | Organic Syntheses | |
| Takai olefination Olefination | Takai olefination in organic chemistry describes the organic reaction of an aldehyde with a diorganochromium compound to form an alkene. It is a name reaction, named for Kazuhiko Takai, who first repo… | Wikipedia: Category:Name reactions | |
| Wittig olefination: methyltriphenylphosphonium bromide + benzaldehyde → styrene Scale Olefination | Canonical Wittig olefination — the textbook 'make an alkene from an aldehyde' demo. The ylide (methylenetriphenylphosphorane) is generated in situ from methyltriphenylphosphonium bromide + base (typic… | Wittig 1954 | |
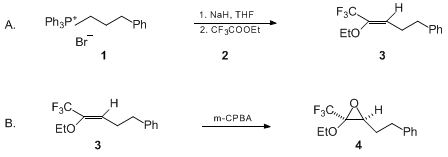
| WITTIG OLEFINATION OF PERFLUOROALKYL CARBOXYLIC ESTERS; SYNTHESIS OF 1,1,1-TRIFLUORO-2-ETHOXY-5-PHENYLPENT-2-ENE AND 1-PERFLUOROALKYL EPOXY ETHERS: 1,1,1-TRIFLUORO-2-ETHOXY-2,3-EPOXY-5-PHENYLPENTANE ((Benzene, (4-ethoxy-5,5,5-trifluoro-3-pentenyl)-, (Z)- and Oxirane, 2-ethoxy-3-(2-phenylethyl)-2-(trifluoromethyl)-, cis-(±)-)) Olefination | Preparation of (Benzene, (4-ethoxy-5,5,5-trifluoro-3-pentenyl)-, (Z)- and Oxirane, 2-ethoxy-3-(2-phenylethyl)-2-(trifluoromethyl)-, cis-(±)-). Submitted by Jean-Pierre Bégué, Danièle Bonnet-Delpon, an… | Organic Syntheses | |
| Wittig reaction Olefination | The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent. Wittig reactions are most commonly used to conve… | Wikipedia: Category:Name reactions |