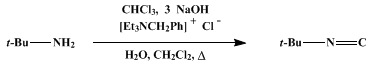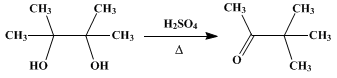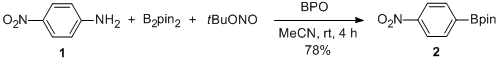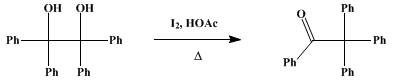Browse · Reactions
Reactions
95 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| Jacobsen rearrangement Rearrangement | The Jacobsen rearrangement is a chemical reaction, commonly described as the migration of an alkyl group in a sulfonic acid derived from a polyalkyl- or polyhalobenzene: | Wikipedia: Category:Name reactions | |
| Kornblum–DeLaMare rearrangement Rearrangement | The Kornblum–DeLaMare rearrangement is a rearrangement reaction in organic chemistry in which a primary or secondary organic peroxide is converted to the corresponding ketone and alcohol under acid or… | Wikipedia: Category:Name reactions | |
| Lossen rearrangement Rearrangement | The Lossen rearrangement is the conversion of an acyl hydroxamate to an isocyanate and carboxylic acid side product. The isocyanate can be used further to generate ureas in the presence of amines or g… | Wikipedia: Category:Name reactions | |
| McLafferty rearrangement Rearrangement | The McLafferty rearrangement is a fragmentation reaction observed in mass spectrometry of organic molecules in which the parent radical cation splits into daughter radical cation and neutral spin-pair… | Wikipedia: Category:Name reactions | |
| Meisenheimer rearrangement Rearrangement | In organic chemistry, the Meisenheimer rearrangement is the conversion of a tertiary amine oxide to a alkoxylamine. The reaction is most common for benzylic or allylic amine oxides. The reaction is … | Wikipedia: Category:Name reactions | |
| METHYL CARBAMATE FORMATION VIA MODIFIED HOFMANN REARRANGEMENT REACTIONS: METHYL N-(p-METHOXYPHENYL)CARBAMATE (Carbamic acid, (4-methoxyphenyl)-, methyl ester) Rearrangement | Preparation of Carbamic acid, (4-methoxyphenyl)-, methyl ester. Submitted by Jeffrey W. Keillor1 and Xicai Huang. DOI: 10.15227/orgsyn.078.0234. | Organic Syntheses | |
| Meyer–Schuster rearrangement Rearrangement | The Meyer–Schuster rearrangement is the chemical reaction described as an acid-catalyzed rearrangement of secondary and tertiary propargyl alcohols to α,β-unsaturated ketones if the alkyne group is in… | Wikipedia: Category:Name reactions | |
| MILD AND EFFICIENT ONE-POT CURTIUS REARRANGEMENT: PREPARATION OF N-tert-BUTYL ADAMANTANYL-1-YL-CARBAMATE Rearrangement | Preparation of MILD AND EFFICIENT ONE-POT CURTIUS REARRANGEMENT: PREPARATION OF N-tert-BUTYL ADAMANTANYL-1-YL-CARBAMATE. Submitted by Olivier Leogane and Hélène Lebel1. DOI: 10.15227/orgsyn.086.0113. | Organic Syntheses | |
| Mislow–Evans rearrangement Rearrangement | The Mislow–Evans rearrangement is a name reaction in organic chemistry. It is named after Kurt Mislow who reported the prototypical reaction in 1966, and David A. Evans who published further developme… | Wikipedia: Category:Name reactions | |
| Mumm rearrangement Rearrangement | The Mumm rearrangement is an organic reaction and a rearrangement reaction. It describes a 1,3(O-N) acyl transfer of an acyl imidate or isoimide group to an imide. | Wikipedia: Category:Name reactions | |
| Neber rearrangement Rearrangement | The Neber rearrangement is an organic reaction in which a ketoxime is converted into an alpha-aminoketone via a rearrangement reaction. | Wikipedia: Category:Name reactions | |
| Newman–Kwart rearrangement Rearrangement | The Newman–Kwart rearrangement is a type of rearrangement reaction in which the aryl group of an O-aryl thiocarbamate, ArOC(=S)NMe2, migrates from the oxygen atom to the sulfur atom, forming an S-aryl… | Wikipedia: Category:Name reactions | |
| Overman rearrangement Rearrangement | The Overman rearrangement is a chemical reaction that can be described as a Claisen rearrangement of allylic alcohols to give allylic trichloroacetamides through an imidate intermediate. The Overman r… | Wikipedia: Category:Name reactions | |
| Oxy-Cope rearrangement Rearrangement | In organic chemistry, the oxy-Cope rearrangement is a chemical reaction. It involves reorganization of the skeleton of certain unsaturated alcohols. It is a variation of the Cope rearrangement in whic… | Wikipedia: Category:Name reactions | |
| Payne rearrangement Rearrangement | The Payne rearrangement is the isomerization, under basic conditions, of 2,3-epoxy alcohols to isomeric 1,2-epoxy alcohols with inversion of configuration. Aza- and thia-Payne rearrangements of azirid… | Wikipedia: Category:Name reactions | |
| PHASE-TRANSFER HOFMANN CARBYLAMINE REACTION: tert-BUTYL ISOCYANIDE (Propane, 2-isocyano-2-methyl-) Rearrangement | Preparation of Propane, 2-isocyano-2-methyl-. Submitted by George W. Gokel1 2, Ronald P. Widera, and William P. Weber1. DOI: 10.15227/orgsyn.055.0096. | Organic Syntheses | |
| Piancatelli rearrangement Rearrangement | In 1976, the Italian chemist, Giovanni Piancatelli and coworkers developed a new method to synthesize 4-hydroxycyclopentenone derivatives from 2-furylcarbinols through an acid-catalyzed rearrangement.… | Wikipedia: Category:Name reactions | |
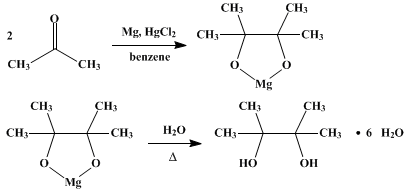
| PINACOL HYDRATE Rearrangement | Preparation of PINACOL HYDRATE. Submitted by Roger Adams and E. W. Adams. DOI: 10.15227/orgsyn.005.0087. | Organic Syntheses | |
| PINACOLONE Rearrangement | Preparation of PINACOLONE. Submitted by G. A. Hill and E. W. Flosdorf. DOI: 10.15227/orgsyn.005.0091. | Organic Syntheses | |
| Pummerer rearrangement Rearrangement | The Pummerer rearrangement is an organic reaction whereby an alkyl sulfoxide rearranges to an α-acyloxy–thioether (monothioacetal-ester) in the presence of acetic anhydride. | Wikipedia: Category:Name reactions | |
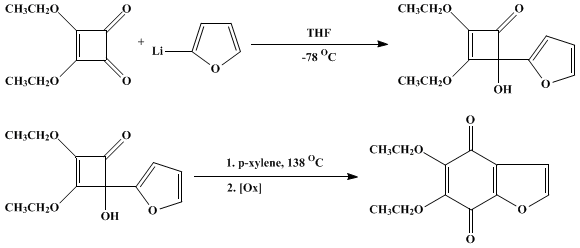
| REARRANGEMENT OF 4-ARYL-4-HYDROXY-2,3-DIALKOXYCYCLOBUTENONES TO ANNULATED HYDROQUINONES AND QUINONES: 5,6-DIETHOXYBENZOFURAN-4,7-DIONE Rearrangement | Preparation of REARRANGEMENT OF 4-ARYL-4-HYDROXY-2,3-DIALKOXYCYCLOBUTENONES TO ANNULATED HYDROQUINONES AND QUINONES: 5,6-DIETHOXYBENZOFURAN-4,7-DIONE. Submitted by S. T. Perri, P. Rice, and H. W. Moor… | Organic Syntheses | |
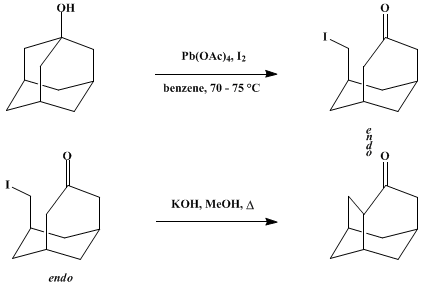
| REARRANGEMENT OF BRIDGEHEAD ALCOHOLS TO POLYCYCLIC KETONES BY FRAGMENTATION-CYCLIZATION: 4-PROTOADAMANTANONE (TRICYCLO-[4.3.1.03,8]DECAN-4-ONE) (2,5-Methano-1H-inden-7(4H)-one, hexahydro) Rearrangement | Preparation of 2,5-Methano-1H-inden-7(4H)-one, hexahydro. Submitted by Zdenko Majerski1 and Zdenko Hamersak1. DOI: 10.15227/orgsyn.059.0147. | Organic Syntheses | |
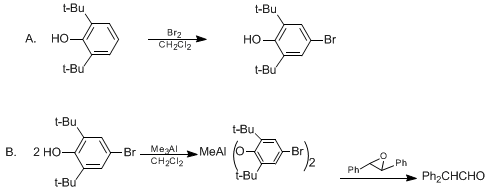
| REARRANGEMENT OF trans-STILBENE OXIDE TO DIPHENYLACETALDEHYDE WITH CATALYTIC METHYLALUMINUM BIS(4-BROMO-2,6-DI-tert-BUTYLPHENOXIDE) (Oxirane, 2,3-diphenyl-, trans- to Benzeneacetaldehyde, α-phenyl-) Rearrangement | Preparation of Oxirane, 2,3-diphenyl-, trans- to Benzeneacetaldehyde, α-phenyl-. Submitted by Takashi Ooi, Keiji Maruoka, and Hisashi Yamamoto1. DOI: 10.15227/orgsyn.072.0095. | Organic Syntheses | |
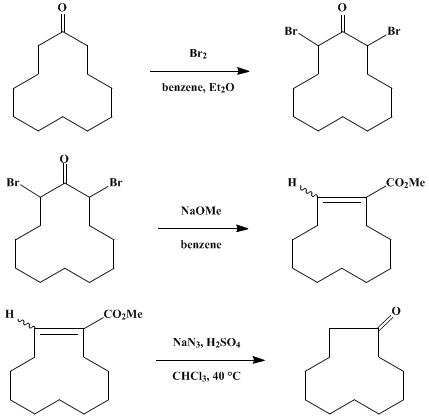
| RING CONTRACTION via A FAVORSKII-TYPE REARRANGEMENT: CYCLOUNDECANONE Rearrangement | Preparation of RING CONTRACTION via A FAVORSKII-TYPE REARRANGEMENT: CYCLOUNDECANONE. Submitted by J. Wohllebe and E. W. Garbisch Jr1. DOI: 10.15227/orgsyn.056.0107. | Organic Syntheses | |
| Schmidt reaction Rearrangement | In organic chemistry, the Schmidt reaction is an organic reaction in which an azide reacts with a carbonyl derivative, usually an aldehyde, ketone, or carboxylic acid, under acidic conditions to give … | Wikipedia: Category:Name reactions | |
| SILVER-CATALYZED REARRANGEMENT OF PROPARGYLIC SULFINATES: SYNTHESIS OF ALLENIC SULFONES Rearrangement | Preparation of SILVER-CATALYZED REARRANGEMENT OF PROPARGYLIC SULFINATES: SYNTHESIS OF ALLENIC SULFONES. Submitted by Michael Harmata, Zhengxin Cai, and Chaofeng Huang1. DOI: 10.15227/orgsyn.088.0309. | Organic Syntheses | |
| Skattebøl rearrangement Rearrangement | The Skattebøl rearrangement is an organic reaction for converting a geminal dihalo cyclopropane to an allene using an organolithium base. This rearrangement reaction is named after its discoverer, Lar… | Wikipedia: Category:Name reactions | |
| Smiles rearrangement Rearrangement | In organic chemistry, the Smiles rearrangement is an organic reaction and a rearrangement reaction named after British chemist Samuel Smiles. It is an intramolecular, nucleophilic aromatic substitutio… | Wikipedia: Category:Name reactions | |
| Sommelet–Hauser rearrangement Rearrangement | The Sommelet–Hauser rearrangement (named after M. Sommelet and Charles R. Hauser) is a rearrangement reaction of certain benzyl quaternary ammonium salts. The reagent is sodium amide or another alkali… | Wikipedia: Category:Name reactions | |
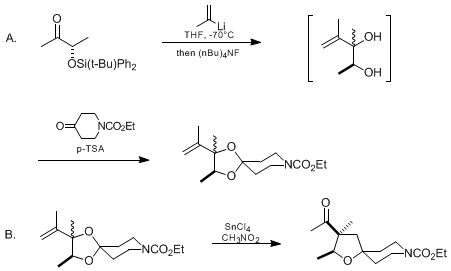
| STEREOCONTROLLED PREPARATION OF 3-ACYLTETRAHYDROFURANS FROM ACID-PROMOTED REARRANGEMENTS OF ALLYLIC KETALS: (2S,3S)-3-ACETYL-8-CARBOETHOXY-2,3-DIMETHYL-1-OXA-8-AZASPIRO[4.5]DECANE Rearrangement | Preparation of STEREOCONTROLLED PREPARATION OF 3-ACYLTETRAHYDROFURANS FROM ACID-PROMOTED REARRANGEMENTS OF ALLYLIC KETALS: (2S,3S)-3-ACETYL-8-CARBOETHOXY-2,3-DIMETHYL-1-OXA-8-AZASPIRO[4.5]DECANE. Subm… | Organic Syntheses | |
| Stevens rearrangement Rearrangement | The Stevens rearrangement in organic chemistry is an organic reaction converting quaternary ammonium salts and sulfonium salts to the corresponding amines or sulfides in presence of a strong base in a… | Wikipedia: Category:Name reactions | |
| Stieglitz rearrangement Rearrangement | The Stieglitz rearrangement is a rearrangement reaction in organic chemistry which is named after the American chemist Julius Stieglitz (1867–1937) and was first investigated by him and Paul Nicholas … | Wikipedia: Category:Name reactions | |
| SYNTHESIS OF AN N-SUBSTITUTED LACTAM USING AN INTRAMOLECULAR SCHMIDT REACTION: FORMATION OF 2,3,11,11a-TETRAHYDRO-1H-BENZO[d]PYRROLO[1,2-a]AZEPIN-5(6H)-ONE Rearrangement | Preparation of SYNTHESIS OF AN N-SUBSTITUTED LACTAM USING AN INTRAMOLECULAR SCHMIDT REACTION: FORMATION OF 2,3,11,11a-TETRAHYDRO-1H-BENZO[d]PYRROLO[1,2-a]AZEPIN-5(6H)-ONE. Submitted by Scott Grecian a… | Organic Syntheses | |
| Synthesis of Arylboronic Pinacol Esters from Corresponding Arylamines Rearrangement | Preparation of Synthesis of Arylboronic Pinacol Esters from Corresponding Arylamines. Submitted by Di Qiu, He Meng, Liang Jin, Shengbo Tang, Shuai Wang, Fangyang Mo, Yan Zhang and Jianbo Wang*1. DOI: … | Organic Syntheses | |
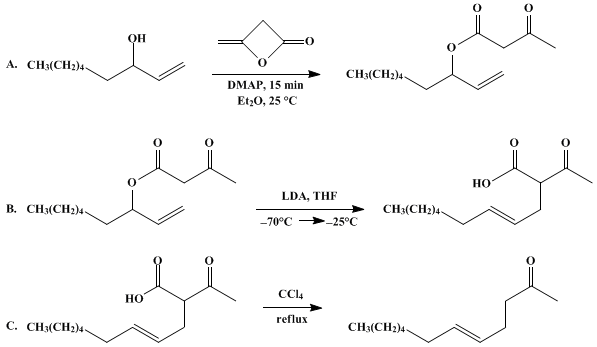
| THE CARROLL REARRANGEMENT: 5-DODECEN-2-ONE (5-Dodecen-2-one, (E)-) Rearrangement | Preparation of 5-Dodecen-2-one, (E)-. Submitted by Stephen R. Wilson and Corinne E. Augelli1. DOI: 10.15227/orgsyn.068.0210. | Organic Syntheses | |
| Tiffeneau–Demjanov rearrangement Rearrangement | The Tiffeneau–Demjanov rearrangement is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone. | Wikipedia: Category:Name reactions | |
| Wagner–Meerwein rearrangement Rearrangement | A Wagner–Meerwein rearrangement is a class of carbocation 1,2-rearrangement reactions in which a hydrogen, alkyl or aryl group migrates from one carbon to a neighboring carbon. | Wikipedia: Category:Name reactions | |
| Wallach rearrangement Rearrangement | The Wallach rearrangement, also named Wallach transformation, is a name reaction in the organic chemistry. It is named after Otto Wallach, who discovered this reaction in 1880. In general it is a stro… | Wikipedia: Category:Name reactions | |
| Westphalen–Lettré rearrangement Rearrangement | The Westphalen–Lettré rearrangement is a classic organic reaction in organic chemistry describing a rearrangement reaction of cholestane-3β,5α,6β-triol diacetate with acetic anhydride and sulfuric aci… | Wikipedia: Category:Name reactions | |
| Willgerodt rearrangement Rearrangement | The Willgerodt rearrangement or Willgerodt reaction is an organic reaction converting an aryl alkyl ketone, alkyne, or alkene to the corresponding amide by reaction with ammonium polysulfide, named af… | Wikipedia: Category:Name reactions | |
| Wolffenstein–Böters reaction Rearrangement | The Wolffenstein–Böters reaction is an organic reaction converting benzene to picric acid by a mixture of aqueous nitric acid and mercury(II) nitrate. | Wikipedia: Category:Name reactions | |
| Wolff–Kishner reduction Rearrangement | The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently emplo… | Wikipedia: Category:Name reactions | |
| Wolff rearrangement Rearrangement | The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearran… | Wikipedia: Category:Name reactions | |
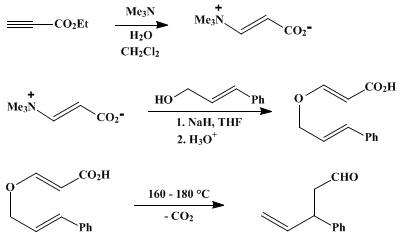
| α-UNSUBSTITUTED γ,δ-UNSATURATED ALDEHYDES BY CLAISEN REARRANGEMENT: 3-PHENYL-4-PENTENAL (Benzenepropanal, β-ethenyl-) Rearrangement | Preparation of Benzenepropanal, β-ethenyl-. Submitted by Dennis E. Vogel and George H. Büchi1. DOI: 10.15227/orgsyn.066.0029. | Organic Syntheses | |
| β-BENZOPINACOLONE Rearrangement | Preparation of β-BENZOPINACOLONE. Submitted by W. E. Bachmann. DOI: 10.15227/orgsyn.014.0012. | Organic Syntheses |