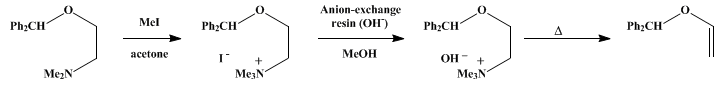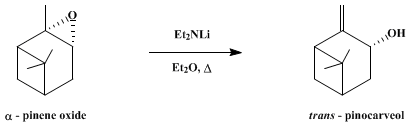Browse · Reactions
Reactions
95 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| 1,2-METALLATE REARRANGEMENT: (Z)-4-(2-PROPENYL)-3-OCTEN-1-OL (3-Octen-1-ol, 4-(2-propenyl)-, (Z)-) Rearrangement | Preparation of 3-Octen-1-ol, 4-(2-propenyl)-, (Z)-. Submitted by Krzysztof Jarowicki, Philip J. Kocienski, and Liu Qun1. DOI: 10.15227/orgsyn.079.0011. | Organic Syntheses | |
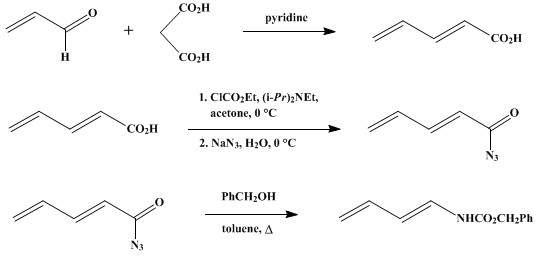
| 1-N-ACYLAMINO-1,3-DIENES FROM 2,4-PENTADIENOIC ACIDS BY THE CURTIUS REARRANGEMENT: BENZYL trans-1,3-BUTADIENE-1-CARBAMATE (Carbamic acid, 1,3-butadienyl-, (E)-, phenylmethyl ester) Rearrangement | Preparation of Carbamic acid, 1,3-butadienyl-, (E)-, phenylmethyl ester. Submitted by Peter J. Jessup, C. Bruce Petty, Jan Roos, and Larry E. Overman1. DOI: 10.15227/orgsyn.059.0001. | Organic Syntheses | |
| ALKENES via HOFMANN ELIMINATION: USE OF ION-EXCHANGE RESIN FOR PREPARATION OF QUATERNARY AMMONIUM HYDROXIDES: DIPHENYLMETHYL VINYL ETHER (Benzene, 1,1'-[(ethenyloxy)methylene]bis-) Rearrangement | Preparation of Benzene, 1,1'-[(ethenyloxy)methylene]bis-. Submitted by Carl Kaiser and Joseph Weinstock1. DOI: 10.15227/orgsyn.055.0003. | Organic Syntheses | |
| Allen–Millar–Trippett rearrangement Rearrangement | The Allen–Millar–Trippett rearrangement is a ring expansion reaction in which a cyclic phosphine is transformed into a cyclic phosphine oxide. This name reaction, first reported in the 1960s by David … | Wikipedia: Category:Name reactions | |
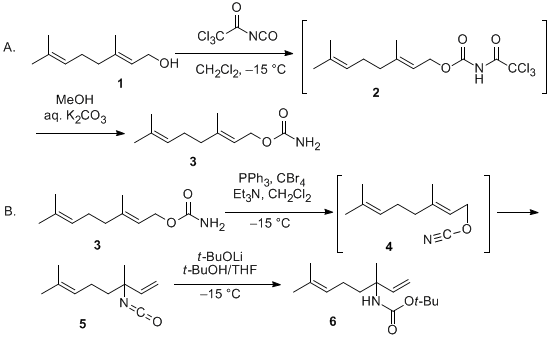
| Allyl Cyanate-To-Isocyanate Rearrangement: Preparation of tert-Butyl 3,7-dimethylocta-1,6-dien-3-ylcarbamate Rearrangement | Preparation of Allyl Cyanate-To-Isocyanate Rearrangement: Preparation of tert-Butyl 3,7-dimethylocta-1,6-dien-3-ylcarbamate. Submitted by Yoshiyasu Ichikawa,1 Noriko Kariya, and Tomoyuki Hasegawa. DOI… | Organic Syntheses | |
| Amadori rearrangement Rearrangement | The Amadori rearrangement is an organic reaction describing the acid or base catalyzed isomerization or rearrangement reaction of the N-glycoside of an aldose or the glycosylamine to the corresponding… | Wikipedia: Category:Name reactions | |
| Aston–Greenburg rearrangement Rearrangement | The Aston–Greenburg rearrangement is a name reaction in organic chemistry. It allows for the generation of tertiary α-alkylesters from corresponding α-haloketones through a 1,2-rearrangement, with the… | Wikipedia: Category:Name reactions | |
| ASYMMETRIC REARRANGEMENT OF ALLYLIC TRICHLOROACETIMIDATES: PREPARATION OF (S)-2,2,2-TRICHLORO-N-(1-PROPYLALLYL)ACETAMIDE (Acetamide, 2,2,2-trichloro-N-[(1S)-1-ethenylbutyl]-) Rearrangement | Preparation of Acetamide, 2,2,2-trichloro-N-[(1S)-1-ethenylbutyl]-. Submitted by Carolyn E. Anderson, Larry E. Overman*, and Mary P. Watson. DOI: 10.15227/orgsyn.082.0134. | Organic Syntheses | |
| Aza-Cope rearrangement Rearrangement | Rearrangements, especially those that can participate in cascade reactions, such as the aza-Cope rearrangements, are of high practical as well as conceptual importance in organic chemistry, due to the… | Wikipedia: Category:Name reactions | |
| Baker–Venkataraman rearrangement Rearrangement | The Baker–Venkataraman rearrangement is the chemical reaction of 2-acetoxyacetophenones with base to form 1,3-diketones. | Wikipedia: Category:Name reactions | |
| Bamberger rearrangement Rearrangement | The Bamberger rearrangement is the chemical reaction of phenylhydroxylamines with strong aqueous acid, which will rearrange to give 4-aminophenols. It is named for the German chemist Eugen Bamberger (… | Wikipedia: Category:Name reactions | |
| BASE-INDUCED REARRANGEMENT OF EPOXIDES TO ALLYLIC ALCOHOLS: trans-PINOCARVEOL (Bicyclo[3.3.1]heptan-3-ol, 6,6-dimethyl-2-methylene-, (1α,3α,5α-) Rearrangement | Preparation of Bicyclo[3.3.1]heptan-3-ol, 6,6-dimethyl-2-methylene-, (1α,3α,5α-. Submitted by J. K. Crandall1 and L. C. Crawley. DOI: 10.15227/orgsyn.053.0017. | Organic Syntheses | |
| Beckmann rearrangement Rearrangement | The Beckmann rearrangement, named after the German chemist Ernst Otto Beckmann (1853–1923), is a rearrangement of an oxime functional group to an amide functional group. The rearrangement has also bee… | Wikipedia: Category:Name reactions | |
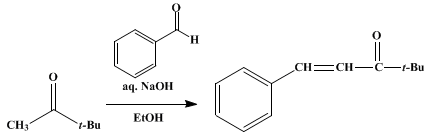
| BENZALPINACOLONE (1-Penten-3-one, 4,4-dimethyl-1-phenyl-) Rearrangement | Preparation of 1-Penten-3-one, 4,4-dimethyl-1-phenyl-. Submitted by G. A. Hill and G. M. Bramann. DOI: 10.15227/orgsyn.005.0015. | Organic Syntheses | |
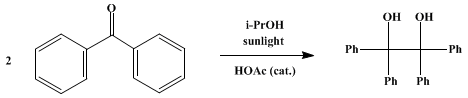
| BENZOPINACOL Rearrangement | Preparation of BENZOPINACOL. Submitted by W. E. Bachmann. DOI: 10.15227/orgsyn.014.0008. | Organic Syntheses | |
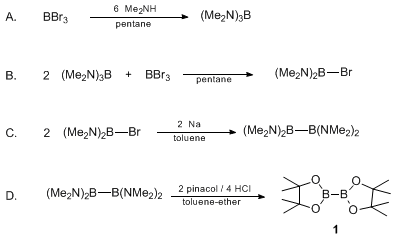
| BIS(PINACOLATO)DIBORON (2,2'-Bi-1,3,2-dioxaborolane, 4,4,4',4',5,5,5',5'-octamethyl-) Rearrangement | Preparation of 2,2'-Bi-1,3,2-dioxaborolane, 4,4,4',4',5,5,5',5'-octamethyl-. Submitted by Tatsuo Ishiyama, Miki Murata, Taka-aki Ahiko, and Norio Miyaura1. DOI: 10.15227/orgsyn.077.0176. | Organic Syntheses | |
| Bohn–Schmidt reaction Rearrangement | The Bohn–Schmidt reaction, a named reaction in chemistry, introduces a hydroxy group at an anthraquinone system. The anthraquinone must already have at least one hydroxy group. The reaction was first … | Wikipedia: Category:Name reactions | |
| Brook rearrangement Rearrangement | In organic chemistry the Brook rearrangement refers to any [1,n] carbon to oxygen silyl migration. The rearrangement was first observed in the late 1950s by Canadian chemist Adrian Gibbs Brook (1924–2… | Wikipedia: Category:Name reactions | |
| Büchner–Curtius–Schlotterbeck reaction Rearrangement | The Buchner–Curtius–Schlotterbeck reaction is the reaction of aldehydes or ketones with aliphatic diazoalkanes to form homologated ketones. It was first described by Eduard Buchner and Theodor Curtius… | Wikipedia: Category:Name reactions | |
| Carroll rearrangement Rearrangement | The Carroll rearrangement is a rearrangement reaction in organic chemistry and involves the transformation of a β-keto allyl ester into a α-allyl-β-ketocarboxylic acid. This organic reaction is accomp… | Wikipedia: Category:Name reactions | |
| Chan rearrangement Rearrangement | The Chan rearrangement is a chemical reaction that involves rearranging an acyloxy acetate (1) in the presence of a strong base to a 2-hydroxy-3-keto-ester (2).[1] | Wikipedia: Category:Name reactions | |
| Chapman rearrangement Rearrangement | The Chapman rearrangement is the thermal conversion of aryl N-arylbenzimidates to the corresponding amides, via intramolecular migration of an aryl group from oxygen to nitrogen. It is named after Art… | Wikipedia: Category:Name reactions | |
| Ciamician–Dennstedt rearrangement Rearrangement | The Ciamician–Dennstedt rearrangement is a name reaction in organic chemistry. It was first published in 1881 by the Italian chemist Giacomo Luigi Ciamician and his German colleague Max Dennstedt. The… | Wikipedia: Category:Name reactions | |
| Claisen condensation Rearrangement | The Claisen condensation is a carbon–carbon bond forming reaction that occurs between two esters or one ester and another carbonyl compound in the presence of a strong base. The reaction produces a β… | Wikipedia: Category:Name reactions | |
| Claisen rearrangement Rearrangement | The Claisen rearrangement is a carbon–carbon bond-forming chemical reaction discovered by Rainer Ludwig Claisen. The heating of an allyl vinyl ether will initiate a [3,3]-sigmatropic rearrangement to … | Wikipedia: Category:Name reactions | |
| Cope reaction Rearrangement | The Cope reaction or Cope elimination, developed by Arthur C. Cope, is the elimination reaction of an N-oxide to an alkene and a hydroxylamine.Typically, the amine oxide is prepared from the correspon… | Wikipedia: Category:Name reactions | |
| Cope rearrangement Rearrangement | The Cope rearrangement (sometimes referred to as the Hardy–Cope rearrangement) is an extensively studied organic reaction involving the [3,3]-sigmatropic rearrangement of 1,5-dienes. It was developed … | Wikipedia: Category:Name reactions | |
| Cornforth rearrangement Rearrangement | In organic chemistry, the Cornforth rearrangement is a rearrangement reaction of a 4-acyloxazole in which the group attached to an acyl on position 4 and the substituent on position 5 of an oxazole ri… | Wikipedia: Category:Name reactions | |
| Criegee rearrangement Rearrangement | The Criegee rearrangement is a rearrangement reaction named after Rudolf Criegee. | Wikipedia: Category:Name reactions | |
| Curtius rearrangement Rearrangement | The Curtius rearrangement (or Curtius reaction or Curtius degradation), first defined by Theodor Curtius in 1885, is the thermal decomposition of an acyl azide to an isocyanate with loss of nitrogen g… | Wikipedia: Category:Name reactions | |
| Demjanov rearrangement Rearrangement | The Demjanov rearrangement is the chemical reaction of primary amines with nitrous acid to give rearranged alcohols.[1][2][3] It involves substitution by a hydroxyl group with a possible ring expansio… | Wikipedia: Category:Name reactions | |
| Dimroth rearrangement Rearrangement | The Dimroth rearrangement is a rearrangement reaction taking place with certain 1,2,3-triazoles where endocyclic and exocyclic nitrogen atoms switch place. This organic reaction was discovered in 1909… | Wikipedia: Category:Name reactions | |
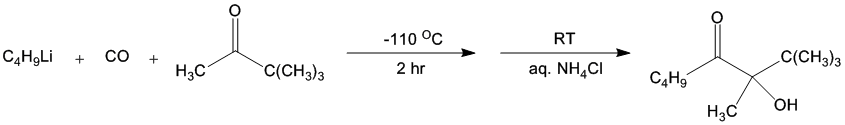
| DIRECT NUCLEOPHILIC ACYLATION BY THE LOW-TEMPERATURE, IN SITU GENERATION OF ACYLLITHIUM REAGENTS; α-HYDROXY KETONES FROM KETONES: 3-HYDROXY-2,2,3-TRIMETHYLOCTAN-4-ONE FROM PINACOLONE (4-Octanone, 3-hydroxy-2,2,3-trimethyl-) Rearrangement | Preparation of 4-Octanone, 3-hydroxy-2,2,3-trimethyl-. Submitted by Richard Hui and Dietmar Seyferth1. DOI: 10.15227/orgsyn.069.0114. | Organic Syntheses | |
| Discussion Addendum for: dl-Selective Pinacol-Type Coupling Using Zinc, Chlorosilane, and Catalytic Amounts of Cp2VCl2; dl-1,2-Dicyclohexylethanediol (1,2-Ethanediol,1,2-dicyclohexyl-) Rearrangement | Preparation of 1,2-Ethanediol,1,2-dicyclohexyl-. Submitted by Toru Amaya and Toshikazu Hirao*1. DOI: 10.15227/orgsyn.089.0073. | Organic Syntheses | |
| dl-SELECTIVE PINACOL-TYPE COUPLING USING ZINC, CHLOROSILANE, AND CATALYTIC AMOUNTS OF Cp2VCl2; dl-1,2-DICYCLOHEXYLETHANEDIOL ((1,2-Ethanediol,1,2-dicyclohexyl-)) Rearrangement | Preparation of (1,2-Ethanediol,1,2-dicyclohexyl-). Submitted by Toshikazu Hirao, Akiya Ogawa, Motoki Asahara, Yasuaki Muguruma, and Hidehiro Sakurai1. DOI: 10.15227/orgsyn.081.0026. | Organic Syntheses | |
| Favorskii rearrangement Rearrangement | In organic chemistry, the Favorskii rearrangement is a reaction of α-halo ketones with a nucleophilic base to acyl derivatives. In the rearrangement, the substrate extrudes the carbonyl carbon to a p… | Wikipedia: Category:Name reactions | |
| Ferrier rearrangement Rearrangement | The Ferrier rearrangement is an organic reaction that involves a nucleophilic substitution reaction combined with an allylic shift in a glycal (a 2,3-unsaturated glycoside). It was discovered by the c… | Wikipedia: Category:Name reactions | |
| Fischer–Hepp rearrangement Rearrangement | In organic chemistry, the Fischer–Hepp rearrangement is a rearrangement reaction in which an aromatic N-nitroso (−N=O) or secondary nitrosamine (>N−N=O) converts to a carbon nitroso compound: | Wikipedia: Category:Name reactions | |
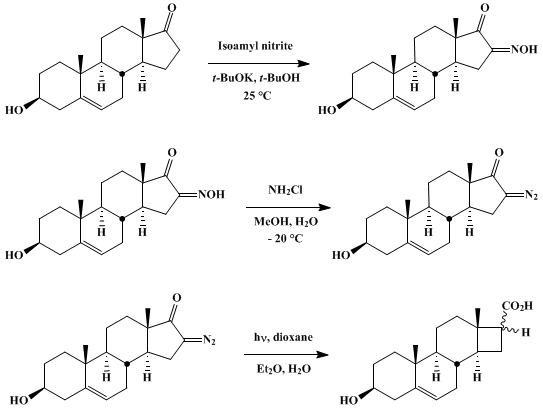
| FORMATION AND PHOTOCHEMICAL WOLFF REARRANGEMENT OF CYCLIC α-DIAZO KETONES: D-NORANDROST-5-EN-3β-OL-16-CARBOXYLIC ACIDS (D-Norandrost-5-ene-16-carboxylic acids, 3β-hydroxy-, (3β,16α) and (3β,16β)-) Rearrangement | Preparation of D-Norandrost-5-ene-16-carboxylic acids, 3β-hydroxy-, (3β,16α) and (3β,16β)-. Submitted by Thomas N. Wheeler and J. Meinwald1. DOI: 10.15227/orgsyn.052.0053. | Organic Syntheses | |
| Fries rearrangement Rearrangement | The Fries rearrangement, named for the German chemist Karl Theophil Fries, is a rearrangement reaction of a phenolic ester to a keto phenol. The reaction is induced by a Lewis acid. | Wikipedia: Category:Name reactions | |
| Fritsch–Buttenberg–Wiechell rearrangement Rearrangement | The Fritsch–Buttenberg–Wiechell rearrangement, named for Paul Ernst Moritz Fritsch (1859–1913), Wilhelm Paul Buttenberg, and Heinrich G. Wiechell, is a chemical reaction whereby a 1,1-diaryl-2-bromo-a… | Wikipedia: Category:Name reactions | |
| Gabriel–Colman rearrangement Rearrangement | The Gabriel–Colman rearrangement is the chemical reaction of a saccharin or phthalimido ester with a strong base, such as an alkoxide, to form substituted isoquinolines. First described in 1900 by che… | Wikipedia: Category:Name reactions | |
| Hayashi rearrangement Rearrangement | The Hayashi rearrangement is the chemical reaction of ortho-benzoylbenzoic acids catalyzed by sulfuric acid or phosphorus pentoxide. | Wikipedia: Category:Name reactions | |
| Hofmann elimination Rearrangement | Hofmann elimination is an elimination reaction of an amine to form alkenes. The least stable alkene (the one with the fewest substituents on the carbons of the double bond), called the Hofmann product… | Wikipedia: Category:Name reactions | |
| Hofmann–Löffler reaction Rearrangement | In organic chemistry, the Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is a radical … | Wikipedia: Category:Name reactions | |
| Hofmann rearrangement Rearrangement | The Hofmann rearrangement (Hofmann degradation) is the organic reaction of a primary amide to a primary amine with one less carbon atom. The reaction involves oxidation of the nitrogen followed by rea… | Wikipedia: Category:Name reactions | |
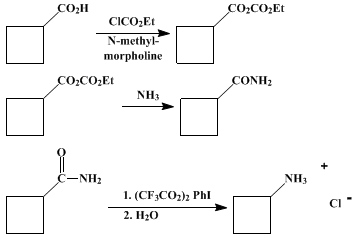
| HOFMANN REARRANGEMENT UNDER MILDLY ACIDIC CONDITIONS USING [I,I-BIS(TRIFLUOROACETOXY)]IODOBENZENE: CYCLOBUTYLAMINE HYDROCHLORIDE FROM CYCLOBUTANECARBOXAMIDE (Cyclobutanamine hydrochloride) Rearrangement | Preparation of Cyclobutanamine hydrochloride. Submitted by Merrick R. Almond, Julie B. Stimmel, E. Alan Thompson, and G. Marc Loudon1. DOI: 10.15227/orgsyn.066.0132. | Organic Syntheses | |
| Homo-Favorskii rearrangement Rearrangement | The homo Favorskii rearrangement is the rearrangement of β-halo ketones and cyclobutanones, which in ring systems may yield ring contraction. This rearrangement takes place in the presence of a base, … | Wikipedia: Category:Name reactions | |
| Intermolecular retro-Cope Type Hydroxylamination of Alkynes with NH2OH: E-1-(1-Hydroxycyclohexyl)ethanone oxime Rearrangement | Preparation of Intermolecular retro-Cope Type Hydroxylamination of Alkynes with NH2OH: E-1-(1-Hydroxycyclohexyl)ethanone oxime. Submitted by Francis Loiseau and André M. Beauchemin1. DOI: 10.15227/org… | Organic Syntheses | |
| Ireland–Claisen rearrangement Rearrangement | The Ireland–Claisen rearrangement is a chemical reaction of an allylic ester with strong base to give an γ,δ-unsaturated carboxylic acid. | Wikipedia: Category:Name reactions |