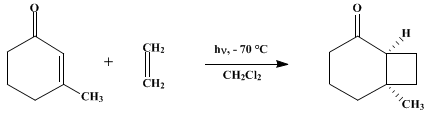Browse · Reactions
Reactions
44 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
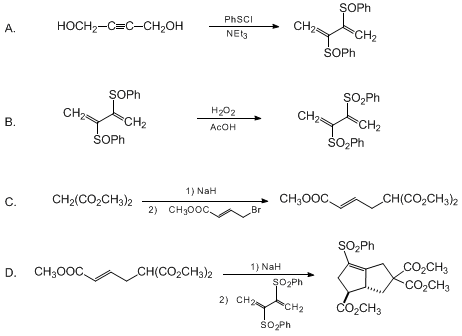
| [3+2]-ANIONIC ELECTROCYCLIZATION USING 2,3-BIS(PHENYLSULFONYL)-1,3-BUTADIENE: trans-4,7,7-TRICARBOMETHOXY-2-PHENYLSULFONYLBICYCLO[3.3.0]OCT-1-ENE (Benzene, 1,1'-[[1,2-bis(methylene)-1,2-ethanediyl]bis(sulfonyl)]bis-) Cyclization | Preparation of Benzene, 1,1'-[[1,2-bis(methylene)-1,2-ethanediyl]bis(sulfonyl)]bis-. Submitted by Albert Padwa1, Scott H. Watterson, and Zhijie Ni. DOI: 10.15227/orgsyn.074.0147. | Organic Syntheses | |
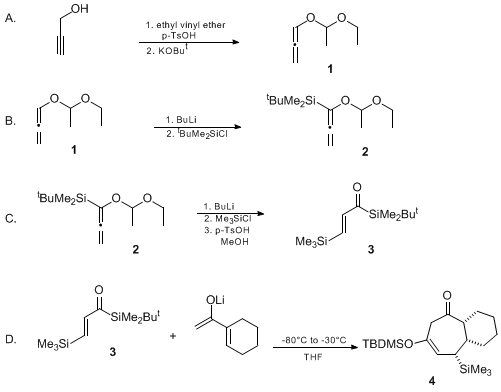
| [3 + 4] ANNULATION USING A [β-(TRIMETHYLSILYL)ACRYLOYL]SILANE AND THE LITHIUM ENOLATE OF AN α,β-UNSATURATED METHYL KETONE: (1R,6S,7S)-4-(tert-BUTYLDIMETHYLSILOXY)-6-(TRIMETHYLSILYL)BICYCLO[5.4.0]UNDEC-4-EN-2-ONE Cyclization | Preparation of [3 + 4] ANNULATION USING A [β-(TRIMETHYLSILYL)ACRYLOYL]SILANE AND THE LITHIUM ENOLATE OF AN α,β-UNSATURATED METHYL KETONE: (1R,6S,7S)-4-(tert-BUTYLDIMETHYLSILOXY)-6-(TRIMETHYLSILYL)BICY… | Organic Syntheses | |
| 5-endo-trig CYCLIZATION OF 1,1-DIFLUORO-1-ALKENES: SYNTHESIS OF 3-BUTYL-2-FLUORO-1-TOSYLINDOLE (1H-Indole, 3-butyl-2-fluoro-1-[(4-methylphenyl)sulfonyl]-) Cyclization | Preparation of 1H-Indole, 3-butyl-2-fluoro-1-[(4-methylphenyl)sulfonyl]-. Submitted by Junji Ichikawa, Ryo Nadano, Takashi Mori, and Yukinori Wada1. DOI: 10.15227/orgsyn.083.0111. | Organic Syntheses | |
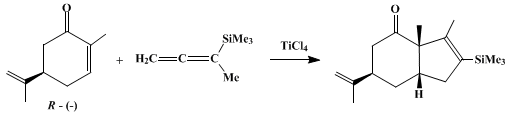
| A GENERAL [3+2] ANNULATION: cis-4-exo-ISOPROPENYL-1,9-DIMETHYL-8-(TRIMETHYLSILYL)BICYCLO[4.3.0]NON-8-EN-2-ONE (4H-Inden-4-one, 1,3a,5,6,7,7a-hexahydro-3,3a-dimethyl-6-(1-methylethenyl)-2-(trimethylsilyl)-, (3aα,6α,7aα)-) Cyclization | Preparation of 4H-Inden-4-one, 1,3a,5,6,7,7a-hexahydro-3,3a-dimethyl-6-(1-methylethenyl)-2-(trimethylsilyl)-, (3aα,6α,7aα)-. Submitted by Rick L. Danheiser, David M. Fink, and Yeun-Min Tsai1. DOI: 10.… | Organic Syntheses | |
| Allan–Robinson reaction Cyclization | The Allan–Robinson reaction is the chemical reaction of o-hydroxyaryl ketones with aromatic anhydrides to form flavones (or isoflavones). | Wikipedia: Category:Name reactions | |
| Bergman cyclization Cyclization | The Masamune-Bergman cyclization or Masamune-Bergman reaction or Masamune-Bergman cycloaromatization is an organic reaction and more specifically a rearrangement reaction taking place when an enediyne… | Wikipedia: Category:Name reactions | |
| Borsche–Drechsel cyclization Cyclization | The Borsche–Drechsel cyclization is a chemical reaction used to synthesize tetrahydrocarbazoles by the acid-catalyzed cyclization of cyclohexanone arylhydrazones. The reaction was first described by E… | Wikipedia: Category:Name reactions | |
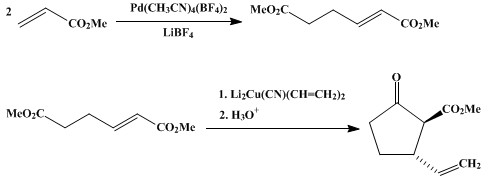
| CONJUGATE ADDITION–CYCLIZATION OF A CYANOCUPRATE: 2-CARBOMETHOXY-3-VINYLCYCLOPENTANONE (Cyclopentanecarboxylic acid, 2-ethenyl-5-oxo-, methyl ester) Cyclization | Preparation of Cyclopentanecarboxylic acid, 2-ethenyl-5-oxo-, methyl ester. Submitted by William A. Nugent and Frank W. Hobbs, Jr.1. DOI: 10.15227/orgsyn.066.0052. | Organic Syntheses | |
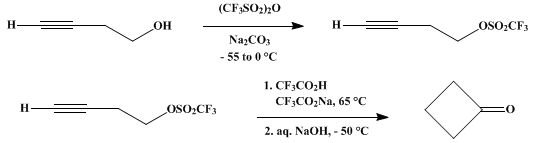
| CYCLOBUTANONE via SOLVOLYTIC CYCLIZATION Cyclization | Preparation of CYCLOBUTANONE via SOLVOLYTIC CYCLIZATION. Submitted by M. Hanack1, T. Dehesch, K. Hummel, and A. Nierth. DOI: 10.15227/orgsyn.054.0084. | Organic Syntheses | |
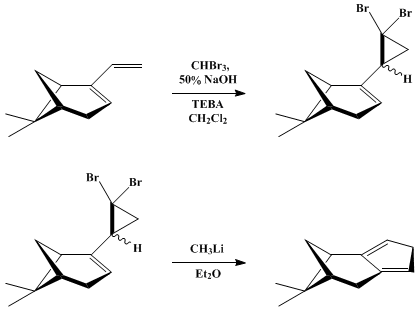
| CYCLOPENTADIENE ANNULATION VIA THE SKATTEBØL REARRANGEMENT: (1R)-9,9-DIMETHYLTRICYCLO-[6.1.1.02,6]DECA-2,5-DIENE Cyclization | Preparation of CYCLOPENTADIENE ANNULATION VIA THE SKATTEBØL REARRANGEMENT: (1R)-9,9-DIMETHYLTRICYCLO-[6.1.1.02,6]DECA-2,5-DIENE. Submitted by Leo A. Paquette and Mark L. McLaughlin1. DOI: 10.15227/org… | Organic Syntheses | |
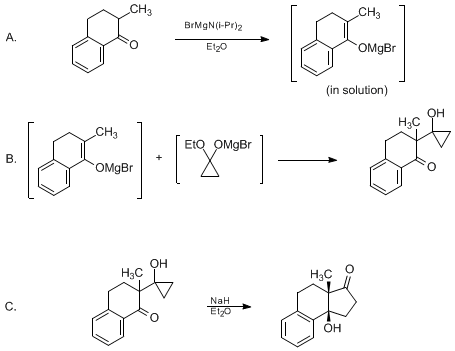
| CYCLOPENTANONE ANNULATION VIA CYCLOPROPANONE DERIVATIVES: (3aβ,9bβ)-1,2,3a,4,5,9b-HEXAHYDRO-9b-HYDROXY-3a-METHYL-3H-BENZ[e]INDEN-3-ONE Cyclization | Preparation of CYCLOPENTANONE ANNULATION VIA CYCLOPROPANONE DERIVATIVES: (3aβ,9bβ)-1,2,3a,4,5,9b-HEXAHYDRO-9b-HYDROXY-3a-METHYL-3H-BENZ[e]INDEN-3-ONE. Submitted by Michael J. Bradlee and Paul Helquist… | Organic Syntheses | |
| Danheiser annulation Cyclization | The Danheiser annulation or Danheiser TMS-cyclopentene annulation is an organic reaction of an α,β-unsaturated ketone and a trialkylsilylallene (e.g., trimethylsilyl- or triisopropylsilyl-) in the pre… | Wikipedia: Category:Name reactions | |
| Danheiser benzannulation Cyclization | The Danheiser benzannulation is a chemical reaction used in organic chemistry to generate highly substituted phenols in a single step. It is named after Rick L. Danheiser who developed the reaction. | Wikipedia: Category:Name reactions | |
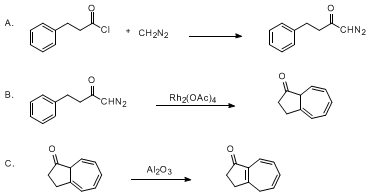
| DIAZO KETONE CYCLIZATION ONTO A BENZENE RING: 3,4-DIHYDRO-1(2H)-AZULENONE (1(2H)-Azulenone, 3,4-dihydro-) Cyclization | Preparation of 1(2H)-Azulenone, 3,4-dihydro-. Submitted by Lawrence T. Scott and Chris A. Sumpter1. DOI: 10.15227/orgsyn.069.0180. | Organic Syntheses | |
| Discussion Addendum for: [3 + 4] Annulation Using a [ß-(Trimethylsilyl) acryloyl]silane and the Lithium Enolate of an a,ß-Unsaturated Methyl Ketone: (1R,6S,7S)-4-(tert-Butyldimethylsiloxy)-6-(trimethylsilyl)bicyclo [5.4.0]undec-4-en-2-one Cyclization | Preparation of Discussion Addendum for: [3 + 4] Annulation Using a [ß-(Trimethylsilyl) acryloyl]silane and the Lithium Enolate of an a,ß-Unsaturated Methyl Ketone: (1R,6S,7S)-4-(tert-Butyldimethylsilo… | Organic Syntheses | |
| Discussion Addendum for: 5-ENDO-TRIG CYCLIZATION OF 1,1-DIFLUORO-1-ALKENES: SYNTHESIS OF 3-BUTYL-2-FLUORO-1-TOSYLINDOLE (1H-INDOLE, 3-BUTYL-2-FLUORO-1-[(4-METHYLPHENYL)SULFONYL]-) Cyclization | Preparation of Discussion Addendum for: 5-ENDO-TRIG CYCLIZATION OF 1,1-DIFLUORO-1-ALKENES: SYNTHESIS OF 3-BUTYL-2-FLUORO-1-TOSYLINDOLE (1H-INDOLE, 3-BUTYL-2-FLUORO-1-[(4-METHYLPHENYL)SULFONYL]-). Subm… | Organic Syntheses | |
| Discussion Addendum for: SYNTHESIS OF INDOLES BY PALLADIUM CATALYZED REDUCTIVE N-HETEROANNULATION OF 2-NITROSTYRENES: METHYL INDOLE-4-CARBOXYLATE Cyclization | Preparation of Discussion Addendum for: SYNTHESIS OF INDOLES BY PALLADIUM CATALYZED REDUCTIVE N-HETEROANNULATION OF 2-NITROSTYRENES: METHYL INDOLE-4-CARBOXYLATE. Submitted by Björn C. Söderberg.*1. DO… | Organic Syntheses | |
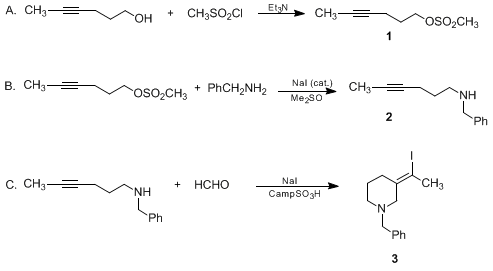
| (E)-1-BENZYL-3-(1-IODOETHYLIDENE)PIPERIDINE: NUCLEOPHILE-PROMOTED ALKYNE-IMINIUM ION CYCLIZATIONS (Piperidine, 3-(iodoethylidene)-1-(phenylmethyl)-(E)-) Cyclization | Preparation of Piperidine, 3-(iodoethylidene)-1-(phenylmethyl)-(E)-. Submitted by H. Arnold, L. E. Overman, M. J. Sharp, and M. C. Witschel1. DOI: 10.15227/orgsyn.070.0111. | Organic Syntheses | |
| ELECTROPHILIC CYCLIZATION WITH N-IODOSUCCINIMIDE: PREPARATION OF 5-(4-BROMOPHENYL)-3-IODO-2-(4-METHYL-PHENYL)FURAN Cyclization | Preparation of ELECTROPHILIC CYCLIZATION WITH N-IODOSUCCINIMIDE: PREPARATION OF 5-(4-BROMOPHENYL)-3-IODO-2-(4-METHYL-PHENYL)FURAN. Submitted by Adam Sniady, Marco S. Morreale, and Roman Dembinski1. DO… | Organic Syntheses | |
| Ferrier carbocyclization Cyclization | The Ferrier carbocyclization (or Ferrier II reaction) is an organic reaction that was first reported by the carbohydrate chemist Robert J. Ferrier in 1979. It is a metal-mediated rearrangement of eno… | Wikipedia: Category:Name reactions | |
| FRAGMENTATION-RECOMBINATION NAZAROV CYCLIZATION: 3,4-DIMETHYLCYCLOPENT-2-EN-1-ONE Cyclization | Preparation of FRAGMENTATION-RECOMBINATION NAZAROV CYCLIZATION: 3,4-DIMETHYLCYCLOPENT-2-EN-1-ONE. Submitted by Keith D. Schwartz and James D. White1. DOI: 10.15227/orgsyn.083.0049. | Organic Syntheses | |
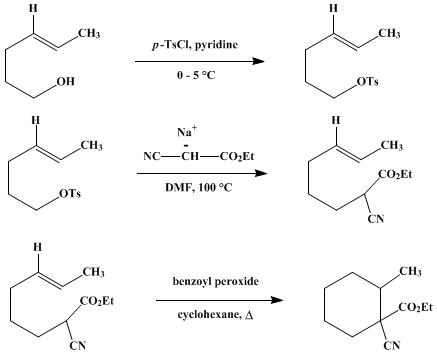
| FREE-RADICAL CYCLIZATION: ETHYL 1-CYANO-2-METHYLCYCLOHEXANECARBOXYLATE (Cyclohexanecarboxylic acid, 1-cyano-2-methyl-, ethyl ester) Cyclization | Preparation of Cyclohexanecarboxylic acid, 1-cyano-2-methyl-, ethyl ester. Submitted by Marc Julia and Michel Maumy1. DOI: 10.15227/orgsyn.055.0057. | Organic Syntheses | |
| GENERATION AND CYCLIZATION OF 5-HEXENYLLITHIUM: 2-CYCLOPENTYLACETOPHENONE (Ethanone, 2-cyclopentyl-1-phenyl-) Cyclization | Preparation of Ethanone, 2-cyclopentyl-1-phenyl-. Submitted by William F. Bailey, Matthew R. Luderer, Michael J. Mealy, and Eric R. Punzalan1. DOI: 10.15227/orgsyn.081.0121. | Organic Syntheses | |
| INDOLE SYNTHESIS BY Pd-CATALYZED ANNULATION OF KETONES WITH o-IODOANILINE: 1,2,3,4-TETRAHYDROCARBAZOLE (1H-Carbazole, 2,3,4,9-tetrahydro-) Cyclization | Preparation of 1H-Carbazole, 2,3,4,9-tetrahydro-. Submitted by Cheng-yi Chen and Robert D. Larsen1. DOI: 10.15227/orgsyn.078.0036. | Organic Syntheses | |
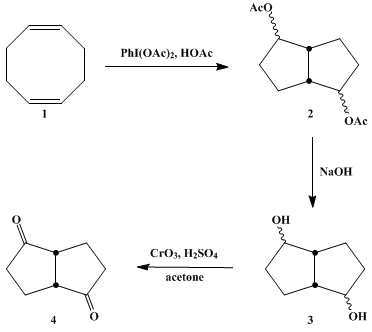
| INTRAMOLECULAR CYCLIZATION OF cis,cis-1,5-CYCLOOCTADIENE USING HYPERVALENT IODINE: BICYCLO[3.3.0]OCTANE-2,6-DIONE (1,4-Pentalenedione, hexahydro-) Cyclization | Preparation of 1,4-Pentalenedione, hexahydro-. Submitted by Robert M. Moriarty1, Michael P. Duncan1, Radhe K. Vaid1, and OM Prakash2. DOI: 10.15227/orgsyn.068.0175. | Organic Syntheses | |
| IRIDIUM-CATALYZED N-HETEROCYCLIZATION OF PRIMARY AMINES WITH DIOLS: N-BENZYLPIPERIDINE Cyclization | Preparation of IRIDIUM-CATALYZED N-HETEROCYCLIZATION OF PRIMARY AMINES WITH DIOLS: N-BENZYLPIPERIDINE. Submitted by Ken-ichi Fujita1a , Youichiro Enoki, and Ryohei Yamaguchi1b. DOI: 10.15227/orgsyn.08… | Organic Syntheses | |
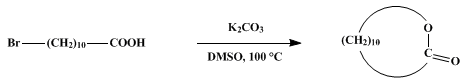
| MACROLIDES FROM CYCLIZATION OF ω-BROMOCARBOXYLIC ACIDS: 11-HYDROXYUNDECANOIC LACTONE (Oxacyclododecan-2-one) Cyclization | Preparation of Oxacyclododecan-2-one. Submitted by C. Galli and L. Mandolini1. DOI: 10.15227/orgsyn.058.0098. | Organic Syntheses | |
| Nazarov cyclization Cyclization | The Nazarov cyclization, also known as the Nazarov reaction, is a chemical reaction used in organic chemistry for the synthesis of cyclopentenones. The reaction is typically divided into classical and… | Wikipedia: Category:Name reactions | |
| PHOSPHINE-CATALYZED [3 + 2] ANNULATION: SYNTHESIS OF ETHYL 5-(tert-BUTYL)-2-PHENYL-1-TOSYL-3-PYRROLINE-3-CARBOXYLATE Cyclization | Preparation of PHOSPHINE-CATALYZED [3 + 2] ANNULATION: SYNTHESIS OF ETHYL 5-(tert-BUTYL)-2-PHENYL-1-TOSYL-3-PYRROLINE-3-CARBOXYLATE. Submitted by Ian P. Andrews and Ohyun Kwon1. DOI: 10.15227/orgsyn.0… | Organic Syntheses | |
| PHOSPHINE-CATALYZED [4+2] ANNULATION: SYNTHESIS OF ETHYL 6-PHENYL-1-TOSYL-1,2,5,6-TETRAHYDROPYRIDINE-3-CARBOXYLATE Cyclization | Preparation of PHOSPHINE-CATALYZED [4+2] ANNULATION: SYNTHESIS OF ETHYL 6-PHENYL-1-TOSYL-1,2,5,6-TETRAHYDROPYRIDINE-3-CARBOXYLATE. Submitted by Kui Lu and Ohyun Kwon1. DOI: 10.15227/orgsyn.086.0212. | Organic Syntheses | |
| PHOTOCYCLIZATION OF AN ENONE TO AN ALKENE: 6-METHYLBICYCLO[4.2.0]OCTAN-2-ONE (Bicyclo[4.2.0]octan-2-one, 6-methyl-) Cyclization | Preparation of Bicyclo[4.2.0]octan-2-one, 6-methyl-. Submitted by R. L. Cargill1, J. R. Dalton, G. H. Morton, and W. E. Caldwell1. DOI: 10.15227/orgsyn.062.0118. | Organic Syntheses | |
| Preparation of 3-Alkylated Oxindoles from N-Benzyl Aniline via a Cu(II)-Mediated Anilide Cyclization Process Cyclization | Preparation of Preparation of 3-Alkylated Oxindoles from N-Benzyl Aniline via a Cu(II)-Mediated Anilide Cyclization Process. Submitted by David S. Pugh1 and Richard J. K. Taylor1. DOI: 10.15227/orgsyn… | Organic Syntheses | |
| Preparation of 5-Hydroxycyclopentenones Via Conjugate Addition-Initiated Nazarov Cyclization Cyclization | Preparation of Preparation of 5-Hydroxycyclopentenones Via Conjugate Addition-Initiated Nazarov Cyclization. Submitted by Joshua L. Brooks, Yu-Wen Huang, and Alison J. Frontier1*. DOI: 10.15227/orgsyn… | Organic Syntheses | |
| Preparation of Tetrahydroisoquinoline-3-ones Via Cyclization of Phenyl Acetamides Using Eaton's Reagent Cyclization | Preparation of Preparation of Tetrahydroisoquinoline-3-ones Via Cyclization of Phenyl Acetamides Using Eaton's Reagent. Submitted by Qiang Yang, Luckner G. Ulysse, Mark D. McLaws, Daniel K. Keefe, Pet… | Organic Syntheses | |
| Pschorr cyclization Cyclization | The Pschorr cyclization is a name reaction in organic chemistry, which was named after its discoverer, the German chemist Robert Pschorr (1868-1930). It describes the intramolecular substitution of ar… | Wikipedia: Category:Name reactions | |
| REDUCTIVE ANNULATION OF VINYL SULFONES: BICYCLO[4.3.0.]NON-1-EN-4-ONE (5H-Inden-5-one, 1,2,3,3a,4,6-hexahydro-) Cyclization | Preparation of 5H-Inden-5-one, 1,2,3,3a,4,6-hexahydro-. Submitted by Ho-shen Lin and Leo A. Paquette1. DOI: 10.15227/orgsyn.067.0163. | Organic Syntheses | |
| Robinson annulation Cyclization | The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson in 1935 as a method to create a six membered ring by forming three new… | Wikipedia: Category:Name reactions | |
| Robinson–Gabriel synthesis Cyclization | The Robinson–Gabriel synthesis is an organic reaction in which a 2-acylamino-ketone reacts intramolecularly followed by a dehydration to give an oxazole. A cyclodehydrating agent is needed to catalyze… | Wikipedia: Category:Name reactions | |
| SYNTHESIS OF 2,3-DISUBSTITUTED INDOLES VIA PALLADIUM-CATALYZED ANNULATION OF INTERNAL ALK YNES: 3-METHYL-2-(TRIMETHYLSILYL)INDOLE Cyclization | Preparation of SYNTHESIS OF 2,3-DISUBSTITUTED INDOLES VIA PALLADIUM-CATALYZED ANNULATION OF INTERNAL ALK YNES: 3-METHYL-2-(TRIMETHYLSILYL)INDOLE. Submitted by Yu Chen, Nataliya A. Markina, Tuanli Yao,… | Organic Syntheses | |
| SYNTHESIS OF Et2SBr•SbCl5Br AND ITS USE IN BIOMIMETIC BROMINATIVE POLYENE CYCLIZATIONS Cyclization | Preparation of SYNTHESIS OF Et2SBr•SbCl5Br AND ITS USE IN BIOMIMETIC BROMINATIVE POLYENE CYCLIZATIONS. Submitted by Scott A. Snyder and Daniel S. Treitler1. DOI: 10.15227/orgsyn.088.0054. | Organic Syntheses | |
| SYNTHESIS OF INDOLES BY PALLADIUM-CATALYZED REDUCTIVE N-HETEROANNULATION OF 2-NITROSTYRENES: METHYL INDOLE-4-CARBOXYLATE ((1H-Indole-4-carboxylic acid, methyl ester)) Cyclization | Preparation of (1H-Indole-4-carboxylic acid, methyl ester). Submitted by Björn C. Söderberg1, James A. Shriver, and Jeffery M. Wallace. DOI: 10.15227/orgsyn.080.0075. | Organic Syntheses | |
| Synthesis of Quinolines by Electrophilic Cyclization of N-(2-Alkynyl)Anilines: 3-Iodo-4-Phenylquinoline Cyclization | Preparation of Synthesis of Quinolines by Electrophilic Cyclization of N-(2-Alkynyl)Anilines: 3-Iodo-4-Phenylquinoline. Submitted by Yu Chen, Anton Dubrovskiy, and Richard C. Larock.1,2. DOI: 10.15227… | Organic Syntheses | |
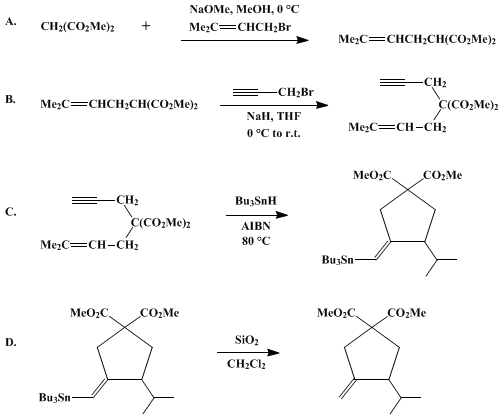
| VINYL RADICAL CYCLIZATION VIA ADDITION OF TIN RADICALS TO TRIPLE BONDS: 3-METHYLENE-4-ISOPROPYL-1,1-CYCLOPENTANEDICARBOXYLIC ACID, DIMETHYL ESTER (1,1-Cyclopentanedicarboxylic acid, 3-methylene-4-(1-methylethyl)-, dimethyl ester) Cyclization | Preparation of 1,1-Cyclopentanedicarboxylic acid, 3-methylene-4-(1-methylethyl)-, dimethyl ester. Submitted by Robert Mook, Jr. and Philip Michael Sher1. DOI: 10.15227/orgsyn.066.0075. | Organic Syntheses | |
| Volhard–Erdmann cyclization Cyclization | The Volhard–Erdmann cyclization is an organic synthesis of alkyl and aryl thiophenes by cyclization of disodium succinate or other 1,4-difunctional compounds (γ-oxo acids, 1,4-diketones, chloroacetyl-… | Wikipedia: Category:Name reactions |