Browse · Reactions
Reactions
58 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| Staudinger reaction Reduction | The Staudinger reaction is a chemical reaction of an organic azide with a phosphine or phosphite produces an iminophosphorane. The reaction was discovered by and named after Hermann Staudinger. The r… | Wikipedia: Category:Name reactions | |
| Staudinger synthesis Reduction | The Staudinger synthesis, also called the Staudinger ketene-imine cycloaddition, is a chemical synthesis in which an imine 1 reacts with a ketene 2 through a non-photochemical 2+2 cycloaddition to pro… | Wikipedia: Category:Name reactions | |
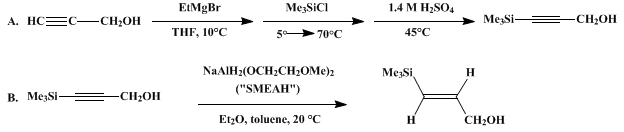
| STEREOSPECIFIC REDUCTION OF PROPARGYL ALCOHOLS: (E)-3-TRIMETHYLSILYL-2-PROPEN-1-OL (2-Propen-1-ol, 3-(trimethylsilyl)-, (E)-) Reduction | Preparation of 2-Propen-1-ol, 3-(trimethylsilyl)-, (E)-. Submitted by Todd K. Jones and Scott E. Denmark1. DOI: 10.15227/orgsyn.064.0182. | Organic Syntheses | |
| SYNTHESIS AND RU(II)-BINAP REDUCTION OF A KETOESTER DERIVED FROM HYDROXYPROLINE: 2(S)-(β-tert-BUTOXYCARBONYL-α-(S) and α-(R)-HYDROXYETHYL)-4(R)-HYDROXYPYRROLIDINE-1-CARBOXYLIC ACID, tert-BUTYL ESTER (2-Pyrrolidinepropanoic acid, 1-[(1,1-dimethylethoxy)carbonyl]-β,4-dihydroxy-, 1,1-dimethylethyl ester, [2S-[2α(S*),4β]]-) Reduction | Preparation of 2-Pyrrolidinepropanoic acid, 1-[(1,1-dimethylethoxy)carbonyl]-β,4-dihydroxy-, 1,1-dimethylethyl ester, [2S-[2α(S*),4β]]-. Submitted by Steven A. King, Joseph Armstrong, and Jennifer Kel… | Organic Syntheses | |
| SYNTHESIS OF 4-TRIISOPROPYLSILYL-3-BUTYN-2-OL BY ASYMMETRIC TRANSFER HYDROGENATION Reduction | Preparation of SYNTHESIS OF 4-TRIISOPROPYLSILYL-3-BUTYN-2-OL BY ASYMMETRIC TRANSFER HYDROGENATION. Submitted by James A. Marshall, Patrick Eidam, and Hilary Schenck Eidam1a. DOI: 10.15227/orgsyn.084.0… | Organic Syntheses | |
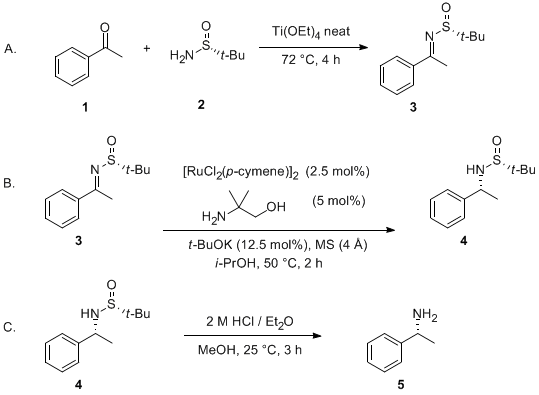
| Synthesis of Highly Enantiomerically Enriched Amines by Asymmetric Transfer Hydrogenation of N-(tert-Butylsulfinyl)Imines Reduction | Preparation of Synthesis of Highly Enantiomerically Enriched Amines by Asymmetric Transfer Hydrogenation of N-(tert-Butylsulfinyl)Imines. Submitted by David Guijarro, Óscar Pablo and Miguel Yus. 1. DO… | Organic Syntheses | |
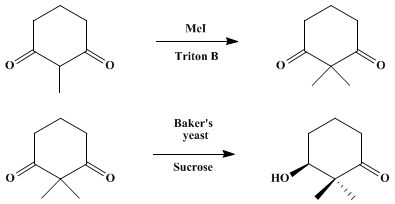
| YEAST REDUCTION OF 2,2-DIMETHYLCYCLOHEXANE-1,3-DIONE: (S)-(+)-3-HYDROXY-2,2-DIMETHYLCYCLOHEXANONE) (Cyclohexanone, 3-hydroxy-2,2-dimethyl-, (S)-) Reduction | Preparation of Cyclohexanone, 3-hydroxy-2,2-dimethyl-, (S)-. Submitted by Kenji Mori and Hideto Mori1. DOI: 10.15227/orgsyn.068.0056. | Organic Syntheses | |
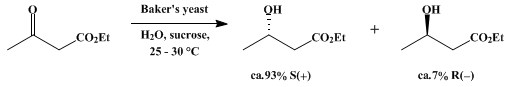
| YEAST REDUCTION OF ETHYL ACETOACETATE: (S)-( + )-ETHYL 3-HYDROXYBUTANOATE (Butanoic acid, 3-hydroxy-, ethyl ester, (S)) Reduction | Preparation of Butanoic acid, 3-hydroxy-, ethyl ester, (S). Submitted by Dieter Seebach, Marius A. Sutter, Roland H. Weber, and Max F. Züger1. DOI: 10.15227/orgsyn.063.0001. | Organic Syntheses |