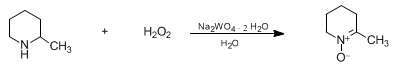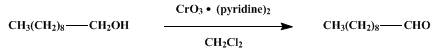Browse · Reactions
Reactions
81 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
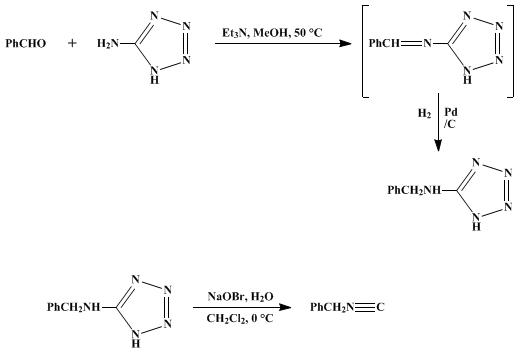
| OXIDATION OF 5-AMINOTETRAZOLES: BENZYL ISOCYANIDE (Benzene, isocyanomethyl) Oxidation | Preparation of Benzene, isocyanomethyl. Submitted by Gerhard Höfle and Bernd Lange1. DOI: 10.15227/orgsyn.061.0014. | Organic Syntheses | |
| OXIDATION OF ALCOHOLS BY METHYL SULFIDE – N-CHLOROSUCCINIMIDE – TRIETHYLAMINE: 4-tert-BUTYLCYCLOHEXANONE (Cyclohexanone, 4-(1,1-dimethylethyl)-) Oxidation | Preparation of Cyclohexanone, 4-(1,1-dimethylethyl)-. Submitted by E. J. Corey1, C. U. Kim2, and P. F. Misco2. DOI: 10.15227/orgsyn.058.0122. | Organic Syntheses | |
| OXIDATION OF NEROL TO NERAL WITH IODOBENZENE DIACETATE AND TEMPO ((Z)-3,7-Dimethyl-2,6-octadienal) Oxidation | Preparation of (Z)-3,7-Dimethyl-2,6-octadienal. Submitted by Giovanni Piancatelli and Francesca Leonelli1a. DOI: 10.15227/orgsyn.083.0018. | Organic Syntheses | |
| OXIDATION OF PRIMARY ALCOHOLS TO CARBOXYLIC ACIDS WITH SODIUM CHLORITE CATALYZED BY TEMPO AND BLEACH: 4-METHOXYPHENYLACETIC ACID ((Benzeneacetic acid, 4-methoxy-)) Oxidation | Preparation of (Benzeneacetic acid, 4-methoxy-). Submitted by Matthew M. Zhao1, Jing Li1, Eiichi Mano2, Zhiguo J. Song1, and David M. Tschaen1. DOI: 10.15227/orgsyn.081.0195. | Organic Syntheses | |
| OXIDATION OF SECONDARY AMINES TO NITRONES: 6-METHYL-2,3,4,5-TETRAHYDROPYRIDINE N-OXIDE (Pyridine, 2,3,4,5-tetrahydro-6-methyl-, 1-oxide) Oxidation | Preparation of Pyridine, 2,3,4,5-tetrahydro-6-methyl-, 1-oxide. Submitted by Shun-Ichi Murahashi, Tatsuki Shiota, and Yasushi Imada1. DOI: 10.15227/orgsyn.070.0265. | Organic Syntheses | |
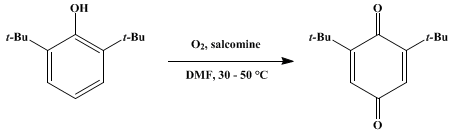
| OXIDATION WITH BIS(SALICYLIDENE)ETHYLENEDIIMINOCOBALT(II) (SALCOMINE): 2,6-DI-tert-BUTYL-p-BENZOQUINONE (2,5-Cyclohexadiene-1,4-dione, 2,6-bis(1,1-dimethylethyl)-) Oxidation | Preparation of 2,5-Cyclohexadiene-1,4-dione, 2,6-bis(1,1-dimethylethyl)-. Submitted by C. R. H. I. De Jonge1, H. J. Hageman, G. Hoentjen, and W. J. Mijs. DOI: 10.15227/orgsyn.057.0078. | Organic Syntheses | |
| OXIDATION WITH THE CHROMIUM TRIOXIDE-PYRIDINE COMPLEX PREPARED in situ:1-DECANAL Oxidation | Preparation of OXIDATION WITH THE CHROMIUM TRIOXIDE-PYRIDINE COMPLEX PREPARED in situ:1-DECANAL. Submitted by R. W. Ratcliffe1. DOI: 10.15227/orgsyn.055.0084. | Organic Syntheses | |
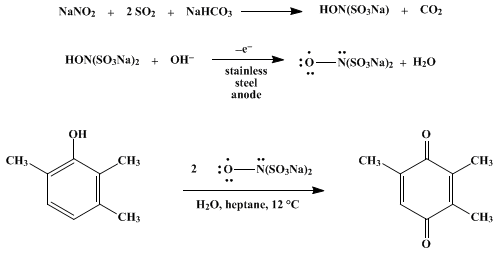
| OXIDATION WITH THE NITROSODISULFONATE RADICAL. I. PREPARATION AND USE OF DISODIUM NITROSODISULFONATE: TRIMETHYL-p-BENZOQUINONE (Nitrosodisulfonic acid, disodium salt and 2,5-Cyclohexadiene-1,4-dione, 2,3,5-trimethyl-) Oxidation | Preparation of Nitrosodisulfonic acid, disodium salt and 2,5-Cyclohexadiene-1,4-dione, 2,3,5-trimethyl-. Submitted by Pius A. Wehrli and Foster Pigott1. DOI: 10.15227/orgsyn.052.0083. | Organic Syntheses | |
| Parikh–Doering oxidation Oxidation | The Parikh–Doering oxidation is an oxidation reaction that transforms primary and secondary alcohols into aldehydes and ketones, respectively. The procedure uses dimethyl sulfoxide (DMSO) as the oxida… | Wikipedia: Category:Name reactions | |
| Pfitzner–Moffatt oxidation Oxidation | The Pfitzner–Moffatt oxidation, sometimes referred to as simply the Moffatt oxidation, is a chemical reaction for the oxidation of primary and secondary alcohols to aldehydes and ketones, respectively… | Wikipedia: Category:Name reactions | |
| Pinnick oxidation Oxidation | The Pinnick oxidation is an organic reaction by which aldehydes can be oxidized into their corresponding carboxylic acids using sodium chlorite (NaClO2) under mild acidic conditions. It was originally… | Wikipedia: Category:Name reactions | |
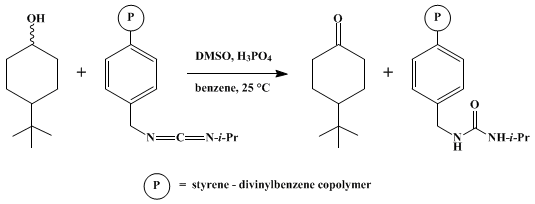
| POLYMERIC CARBODIIMIDE. MOFFAT OXIDATION: 4-tert-BUTYLCYCLOHEXANONE (Cyclohexanone, 4-(1,1-dimethylethyl)-) Oxidation | Preparation of Cyclohexanone, 4-(1,1-dimethylethyl)-. Submitted by Ned M. Weinshenker1, Chah M. Shen, and Jack Y. Wong. DOI: 10.15227/orgsyn.056.0099. | Organic Syntheses | |
| PREPARATION OF 4-ACETYLAMINO-2,2,6,6-TETRAMETHYLPIPERIDINE-1-OXOAMMONIUM TETRAFLUOROBORATE, AND THE OXIDATION OF GERANIOL TO GERANIAL (2,6-Octadienal, 3,7-dimethyl-, (2E)-) Oxidation | Preparation of 2,6-Octadienal, 3,7-dimethyl-, (2E)-. Submitted by James M. Bobbitt and Nabyl Merbouh1. DOI: 10.15227/orgsyn.082.0080. | Organic Syntheses | |
| PREPARATION OF CHIRAL, NONRACEMIC γ-LACTONES BY ENZYMECATALYZED OXIDATION OF meso-DIOLS: ( + )-(1R,6S)-8-OXABICYCLO[4.3.0]NONAN-7-ONE (1(3H)-Isobenzofuranone, hexahydro-, (3aS-cis)-) Oxidation | Preparation of 1(3H)-Isobenzofuranone, hexahydro-, (3aS-cis)-. Submitted by J. Bryan Jones and Ignac J. Jakovac1. DOI: 10.15227/orgsyn.063.0010. | Organic Syntheses | |
| Riley oxidation Oxidation | The Riley oxidation is a selenium dioxide-mediated oxidation of methylene groups adjacent to carbonyls. It was first reported by Harry Lister Riley and co-workers in 1932. In the decade that ensued, s… | Wikipedia: Category:Name reactions | |
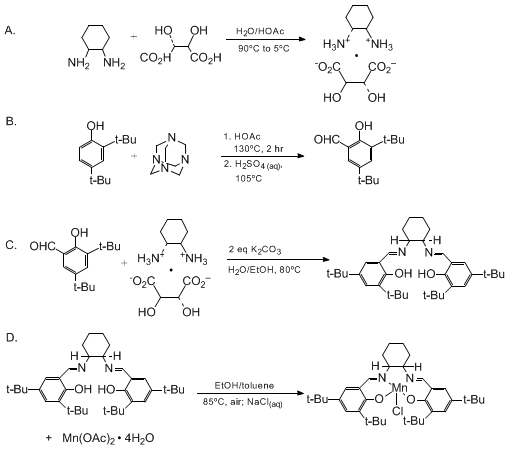
| (R,R)-N,N'-BIS(3,5-DI-tert-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO MANGANESE(III) CHLORIDE, A HIGHLY ENANTIOSELECTIVE EPOXIDATION CATALYST (Manganese, chloro[[2,2'-[1,2-cyclohexanediylbis(nitrilomethylidyne)]-bis[4,6-bis(1,1-dimethylethyl)phenalato]](2-)-N,N',O,O']-, [SP-5-13-(1R-trans-]-) Oxidation | Preparation of Manganese, chloro[[2,2'-[1,2-cyclohexanediylbis(nitrilomethylidyne)]-bis[4,6-bis(1,1-dimethylethyl)phenalato]](2-)-N,N',O,O']-, [SP-5-13-(1R-trans-]-. Submitted by Jay F. Larrow and Eri… | Organic Syntheses | |
| Rubottom oxidation Oxidation | The Rubottom oxidation is a useful, high-yielding chemical reaction between silyl enol ethers and peroxyacids to give the corresponding α-hydroxy carbonyl product. The mechanism of the reaction was … | Wikipedia: Category:Name reactions | |
| Saegusa–Ito oxidation Oxidation | The Saegusa–Ito oxidation is a chemical reaction used in organic chemistry. It was discovered in 1978 by Takeo Saegusa and Yoshihiko Ito as a method to introduce α-β unsaturation in carbonyl compounds… | Wikipedia: Category:Name reactions | |
| Sarett oxidation Oxidation | The Sarett oxidation is an organic reaction that oxidizes primary and secondary alcohols to aldehydes and ketones, respectively, using chromium trioxide and pyridine. Unlike the similar Jones oxidatio… | Wikipedia: Category:Name reactions | |
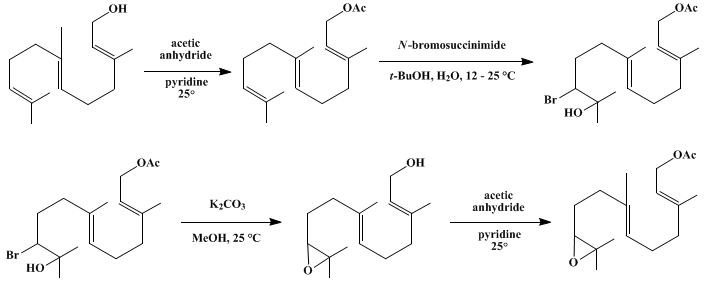
| SELECTIVE EPOXIDATION OF TERMINAL DOUBLE BONDS: 10,11-EPOXYFARNESYL ACETATE (2,6-Nonadien-1-ol, 9-(3,3-dimethyloxiranyl)-3,7-dimethyl-, acetate, (E,E)-) Oxidation | Preparation of 2,6-Nonadien-1-ol, 9-(3,3-dimethyloxiranyl)-3,7-dimethyl-, acetate, (E,E)-. Submitted by R. P. Hanzlik1. DOI: 10.15227/orgsyn.056.0112. | Organic Syntheses | |
| Sharpless epoxidation Oxidation | The Sharpless epoxidation reaction is an enantioselective chemical reaction to prepare 2,3-epoxyalcohols from primary and secondary allylic alcohols. The oxidizing agent is tert-butyl hydroperoxide. … | Wikipedia: Category:Name reactions | |
| Shi epoxidation Oxidation | The Shi epoxidation is a chemical reaction, as the asymmetric epoxidation of alkenes with oxone (potassium peroxymonosulfate) and a fructose-derived catalyst (1). This reaction is thought to proceed v… | Wikipedia: Category:Name reactions | |
| Stahl oxidation Oxidation | The Stahl oxidation is a copper-catalyzed aerobic oxidation of primary and secondary alcohols to aldehydes and ketones. Known for its high selectivity and mild reaction conditions, the Stahl oxidation… | Wikipedia: Category:Name reactions | |
| Swern oxidation Oxidation | In organic chemistry, the Swern oxidation also known as Moffatt–Swern, named after Daniel Swern, is a chemical reaction whereby a primary or secondary alcohol (−OH) is oxidized to an aldehyde (−CH=O) … | Wikipedia: Category:Name reactions | |
| SYNTHESIS OF 1,1-DIMETHYLETHYL (S)-4-FORMYL-2,2-DIMETHYL-3-OXAZOLIDINECARBOXYLATE BY OXIDATION OF THE ALCOHOL (3-Oxazolidinecarboxylic acid, 4-formyl-2,2-dimethyl-, 1,1-dimethylethyl ester, (S)-) Oxidation | Preparation of 3-Oxazolidinecarboxylic acid, 4-formyl-2,2-dimethyl-, 1,1-dimethylethyl ester, (S)-. Submitted by Alessandro Dondoni and Daniela Perrone1. DOI: 10.15227/orgsyn.077.0064. | Organic Syntheses | |
| SYNTHESIS OF 1,2:4,5-DI-O-ISOPROPYLIDENE-D-erythro-2,3-HEXODIULO-2,6-PYRANOSE. A HIGHLY ENANTIOSELECTIVE KETONE CATALYST FOR EPOXIDATION ([β-D-erythro-2,3-Hexodiulo-2,6-pyranose, 1,2:4,5-bis-O-(1-methylethylidene)-]) Oxidation | Preparation of [β-D-erythro-2,3-Hexodiulo-2,6-pyranose, 1,2:4,5-bis-O-(1-methylethylidene)-]. Submitted by Yong Tu, Michael Frohn, Zhi-Xian Wang, and Yian Shi.1. DOI: 10.15227/orgsyn.080.0001. | Organic Syntheses | |
| Synthesis of 1-Naphthol via Oxidation of Potassium 1-Naphthyltrifluoroborate Oxidation | Preparation of Synthesis of 1-Naphthol via Oxidation of Potassium 1-Naphthyltrifluoroborate. Submitted by Gary A. Molander, Sabahat Zahra Siddiqui, and Nicolas Fleury-Brégeot1. DOI: 10.15227/orgsyn.09… | Organic Syntheses | |
| TETRAHYDRO-3-BENZAZEPIN-2-ONES BY LEAD TETRAACETATE OXIDATION OF ISOQUINOLINE ENAMIDES: 7,8-DIMETHOXY-1,3,4,5-TETRAHYDRO-2H-3-BENZAZEPIN-2-ONE (2H-3-Benzazepin-2-one, 1,3,4,5-tetrahydro-7,8-dimethoxy-) Oxidation | Preparation of 2H-3-Benzazepin-2-one, 1,3,4,5-tetrahydro-7,8-dimethoxy-. Submitted by George R. Lenz and Ralph A. Lessor1. DOI: 10.15227/orgsyn.070.0139. | Organic Syntheses | |
| THE DESS-MARTIN PERIODINANE: 1,1,1-TRIACETOXY-1,1-DIHYDRO-1,2-BENZIODOXOL-3(1H)-ONE (1,2-Benziodoxol-3(1H)-one, 1,1,1-tris(acetyloxy)-1,1-dihydro-) Oxidation | Preparation of 1,2-Benziodoxol-3(1H)-one, 1,1,1-tris(acetyloxy)-1,1-dihydro-. Submitted by Robert K. Boeckman, Jr., Pengcheng Shao, and Joseph J. Mullins1. DOI: 10.15227/orgsyn.077.0141. | Organic Syntheses | |
| (±) trans-3,3'-(1,2-CYCLOPROPANEDIYL)BIS-2-(E)-PROPENOIC ACID, DIETHYL ESTER : TANDEM OXIDATION procedure (TOP) USING MnO2 OXIDATION-STABILIZED PHOSPHORANE TRAPPING Oxidation | Preparation of (±) trans-3,3'-(1,2-CYCLOPROPANEDIYL)BIS-2-(E)-PROPENOIC ACID, DIETHYL ESTER : TANDEM OXIDATION procedure (TOP) USING MnO2 OXIDATION-STABILIZED PHOSPHORANE TRAPPING. Submitted by Richar… | Organic Syntheses | |
| α-HYDROXY KETONES FROM THE OXIDATION OF ENOL SILYL ETHERS WITH m-CHLOROPERBENZOIC ACID: 6-HYDROXY- 3,5,5-TRIMETHYL-2-CYCLOHEXEN-1-ONE (2-Cyclohexen-1-one, 6-hydroxy-3,5,5-trimethyl-) Oxidation | Preparation of 2-Cyclohexen-1-one, 6-hydroxy-3,5,5-trimethyl-. Submitted by George M. Rubottom, John M. Gruber, Henrik D. Juve, Jr., and Dan A. Charleson1. DOI: 10.15227/orgsyn.064.0118. | Organic Syntheses |