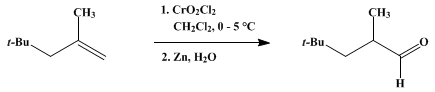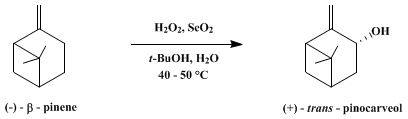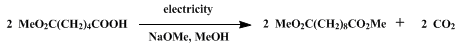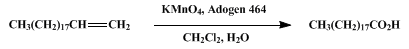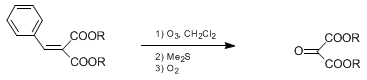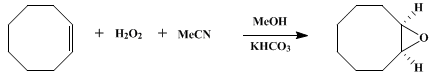Browse · Reactions
Reactions
81 reactions imported from Wikipedia named-reactions and Organic Syntheses archives. Filter by source or search by name + summary.
Results
| Equation | Name | Summary | Source |
|---|---|---|---|
| 2-Iodoxy-5-Methylbenzenesulfonic Acid-Catalyzed Selective Oxidation of 4-Bromobenzyl Alcohol to 4-Bromobenzaldehyde or 4-Bromobenzoic Acid with Oxone Oxidation | Preparation of 2-Iodoxy-5-Methylbenzenesulfonic Acid-Catalyzed Selective Oxidation of 4-Bromobenzyl Alcohol to 4-Bromobenzaldehyde or 4-Bromobenzoic Acid with Oxone. Submitted by Muhammet Uyanik and K… | Organic Syntheses | |
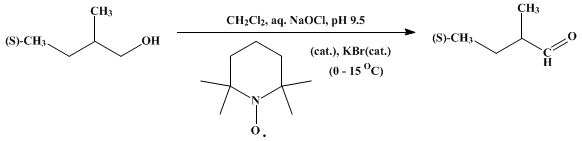
| A GENERAL SYNTHETIC METHOD FOR THE OXIDATION OF PRIMARY ALCOHOLS TO ALDEHYDES: (S)-(+)-2-METHYLBUTANAL (Butanal, 2-methyl-, (S)-) Oxidation | Preparation of Butanal, 2-methyl-, (S)-. Submitted by Pier Lucio Anelli, Fernando Montanari, and Silvio Quici1. DOI: 10.15227/orgsyn.069.0212. | Organic Syntheses | |
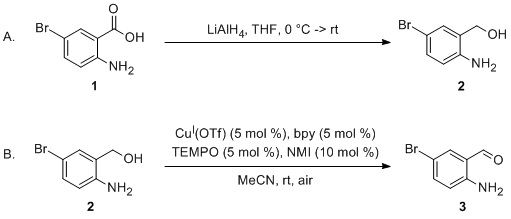
| Air Oxidation of Primary Alcohols Catalyzed by Copper(I)/TEMPO. Preparation of 2-Amino-5-bromobenzaldehyde Oxidation | Preparation of Air Oxidation of Primary Alcohols Catalyzed by Copper(I)/TEMPO. Preparation of 2-Amino-5-bromobenzaldehyde. Submitted by Jessica M. Hoover and Shannon S. Stahl. 1. DOI: 10.15227/orgsyn.… | Organic Syntheses | |
| Albright–Goldman oxidation Oxidation | The Albright–Goldman oxidation is a name reaction of organic chemistry, first described by the American chemists J. Donald Albright and Leon Goldman in 1965. The reaction is particularly suitable for … | Wikipedia: Category:Name reactions | |
| ALDEHYDES BY OXIDATION OF TERMINAL OLEFINS WITH CHROMYL CHLORIDE: 2,4,4-TRIMETHYLPENTANAL (Pentanal, 2,4,4-trimethyl-) Oxidation | Preparation of Pentanal, 2,4,4-trimethyl-. Submitted by Fillmore Freeman1, Richard H. DuBois, and Thomas G. McLaughlin. DOI: 10.15227/orgsyn.051.0004. | Organic Syntheses | |
| ALDEHYDES FROM PRIMARY ALCOHOLS BY OXIDATION WITH CHROMIUM TRIOXIDE: HEPTANAL Oxidation | Preparation of ALDEHYDES FROM PRIMARY ALCOHOLS BY OXIDATION WITH CHROMIUM TRIOXIDE: HEPTANAL. Submitted by J. C. Collins1 and W. W. Hess2. DOI: 10.15227/orgsyn.052.0005. | Organic Syntheses | |
| Allylic Oxidation Catalyzed by Dirhodium(II) Tetrakis[ε-caprolactamate] of tert-Butyldimethylsilyl-protected trans-Dehydroandrosterone Oxidation | Preparation of Allylic Oxidation Catalyzed by Dirhodium(II) Tetrakis[ε-caprolactamate] of tert-Butyldimethylsilyl-protected trans-Dehydroandrosterone. Submitted by Maxim O. Ratnikov1*, Petra L. Goldma… | Organic Syntheses | |
| ALLYLIC OXIDATION WITH HYDROGEN PEROXIDE–SELENIUM DIOXIDE: trans-PINOCARVEOL (Bicyclo[3.1.1]heptan-3-ol, 6,6-dimethyl-2-methylene-, (1α,3α,5α)-) Oxidation | Preparation of Bicyclo[3.1.1]heptan-3-ol, 6,6-dimethyl-2-methylene-, (1α,3α,5α)-. Submitted by J. M. Coxon, E. Dansted, and M. P. Hartshorn1. DOI: 10.15227/orgsyn.056.0025. | Organic Syntheses | |
| ANODIC OXIDATION OF ACIDS: DIMETHYL DECANEDIOATE (Decanedioic acid, dimethyl ester) Oxidation | Preparation of Decanedioic acid, dimethyl ester. Submitted by D. A. White1. DOI: 10.15227/orgsyn.060.0001. | Organic Syntheses | |
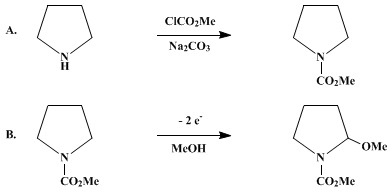
| ANODIC OXIDATION OF N-CARBOMETHOXYPYRROLIDINE: 2-METHOXY-N-CARBOMETHOXYPYRROLIDINE (1-Pyrrolidinecarboxylic acid, 2-methoxy-, methyl ester) Oxidation | Preparation of 1-Pyrrolidinecarboxylic acid, 2-methoxy-, methyl ester. Submitted by T. Shono, Y. Matsumura, and K. Tsubata1. DOI: 10.15227/orgsyn.063.0206. | Organic Syntheses | |
| A PRACTICAL PROCEDURE FOR CARBONYL α-OXIDATION: SYNTHESIS OF (2-BENZOYLOXY)-1,4-CYCLOHEXANEDIONE MONO-ETHYLENE KETAL Oxidation | Preparation of A PRACTICAL PROCEDURE FOR CARBONYL α-OXIDATION: SYNTHESIS OF (2-BENZOYLOXY)-1,4-CYCLOHEXANEDIONE MONO-ETHYLENE KETAL. Submitted by Teyrnon C. Jones and Nicholas C. O. Tomkinson1. DOI: 1… | Organic Syntheses | |
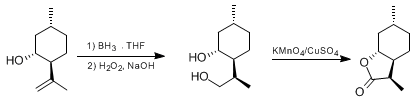
| A SELECTIVE, HETEROGENEOUS OXIDATION USING A MIXTURE OF POTASSIUM PERMANGANATE AND CUPRIC SULFATE: (3aS,7aR)-HEXAHYDRO-(3S,6R)-DIMETHYL-2(3H)-BENZOFURANONE (2(3H)-Benzofuranone, hexahydro-3,6-dimethyl-, [3R-(3α,3aβ,6β,7aα)]-) Oxidation | Preparation of 2(3H)-Benzofuranone, hexahydro-3,6-dimethyl-, [3R-(3α,3aβ,6β,7aα)]-. Submitted by Charles W. Jefford, Yun Li, and Ying Wang1. DOI: 10.15227/orgsyn.071.0207. | Organic Syntheses | |
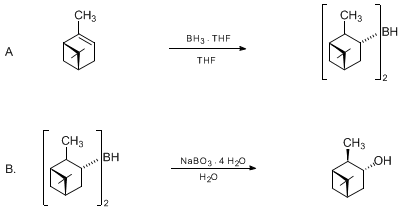
| A SIMPLE AND CONVENIENT METHOD FOR THE OXIDATION OF ORGANOBORANES USING SODIUM PERBORATE: (+)-ISOPINOCAMPHEOL (Bicyclo[3.1.1]heptan-3-ol, 2,6,6-trimethyl-, [1S-(1α,2β,3α,5α)]-) Oxidation | Preparation of Bicyclo[3.1.1]heptan-3-ol, 2,6,6-trimethyl-, [1S-(1α,2β,3α,5α)]-. Submitted by George W. Kabalka, John T. Maddox, Timothy Shoup, and Karla R. Bowers1. DOI: 10.15227/orgsyn.073.0116. | Organic Syntheses | |
| ASYMMETRIC EPOXIDATION OF trans-β-METHYLSTYRENE AND 1-PHENYLCYCLOHEXENE USING A D-FRUCTOSE-DERIVED KETONE: (R,R)-trans-β-METHYLSTYRENE OXIDE AND (R,R)-1-PHENYLCYCLOHEXENE OXIDE ((Oxirane, 2-methyl-3-phenyl-, (2R,3R)- and 7-Oxabicyclo[4.1.0]heptane,1-phenyl-)) Oxidation | Preparation of (Oxirane, 2-methyl-3-phenyl-, (2R,3R)- and 7-Oxabicyclo[4.1.0]heptane,1-phenyl-). Submitted by Zhi-Xian Wang, Lianhe Shu, Michael Frohn, Yong Tu, and Yian Shi1. DOI: 10.15227/orgsyn.080… | Organic Syntheses | |
| Babler oxidation Oxidation | The Babler oxidation, also known as the Babler–Dauben oxidation, is an organic reaction for the oxidative transposition of tertiary allylic alcohols to enones using pyridinium chlorochromate (PCC): | Wikipedia: Category:Name reactions | |
| Baeyer–Villiger oxidation Oxidation | The Baeyer–Villiger oxidation is an organic reaction that forms an ester from a ketone or a lactone from a cyclic ketone, using peroxyacids or peroxides as the oxidant. The reaction is named after Ado… | Wikipedia: Category:Name reactions | |
| Boyland–Sims oxidation Oxidation | The Boyland–Sims oxidation is the chemical reaction of anilines with alkaline potassium persulfate, which after hydrolysis forms ortho-hydroxyl anilines. The reaction is generally performed in water a… | Wikipedia: Category:Name reactions | |
| CARBOXYLIC ACIDS FROM THE OXIDATION OF TERMINAL ALKENES BY PERMANGANATE: NONADECANOIC ACID Oxidation | Preparation of CARBOXYLIC ACIDS FROM THE OXIDATION OF TERMINAL ALKENES BY PERMANGANATE: NONADECANOIC ACID. Submitted by Donald G. Lee, Shannon E. Lamb, and Victor S. Chang1. DOI: 10.15227/orgsyn.060.0… | Organic Syntheses | |
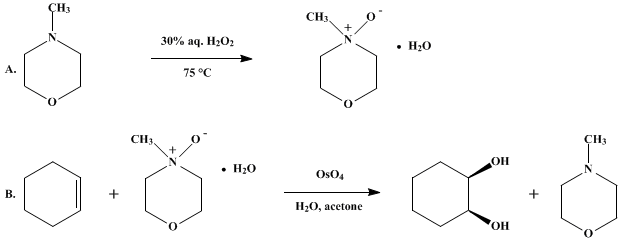
| CATALYTIC OSMIUM TETROXIDE OXIDATION OF OLEFINS: cis-1,2-CYCLOHEXANEDIOL Oxidation | Preparation of CATALYTIC OSMIUM TETROXIDE OXIDATION OF OLEFINS: cis-1,2-CYCLOHEXANEDIOL. Submitted by V. VanRheenen, D. Y. Cha, and W. M. Hartley1. DOI: 10.15227/orgsyn.058.0043. | Organic Syntheses | |
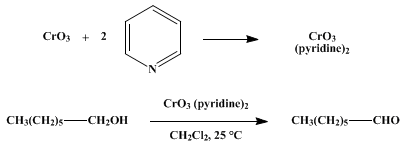
| Collins oxidation Oxidation | The Collins oxidation is an organic reaction for the oxidation of primary alcohols to aldehydes. It is distinguished from other chromium oxide-based oxidations by the use of Collins reagent, a comple… | Wikipedia: Category:Name reactions | |
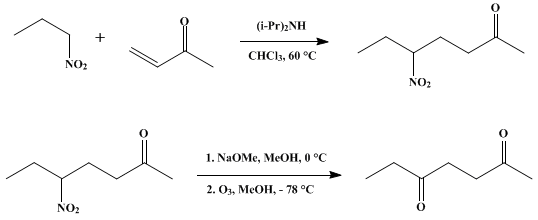
| CONVERSION OF NITRO TO CARBONYL BY OZONOLYSIS OF NITRONATES: 2,5-HEPTANEDIONE Oxidation | Preparation of CONVERSION OF NITRO TO CARBONYL BY OZONOLYSIS OF NITRONATES: 2,5-HEPTANEDIONE. Submitted by John E. McMurry1 and Jack Melton. DOI: 10.15227/orgsyn.056.0036. | Organic Syntheses | |
| Corey–Kim oxidation Oxidation | The Corey–Kim oxidation is an oxidation reaction used to synthesize aldehydes and ketones from primary and secondary alcohols. It is named for American chemist and Nobel Laureate Elias James Corey and… | Wikipedia: Category:Name reactions | |
| Criegee oxidation Oxidation | The Criegee oxidation is a glycol cleavage reaction in which vicinal diols are oxidized to form ketones and aldehydes using lead tetraacetate. It is analogous to the use of periodate (Malaprade react… | Wikipedia: Category:Name reactions | |
| Dakin oxidation Oxidation | The Dakin oxidation (or Dakin reaction) is an organic redox reaction in which an ortho- or para-hydroxylated phenyl aldehyde (2-hydroxybenzaldehyde or 4-hydroxybenzaldehyde) or ketone reacts with hyd… | Wikipedia: Category:Name reactions | |
| Davis oxidation Oxidation | In organic chemistry, the Davis oxidation or Davis' oxaziridine oxidation refers to oxidations involving the use of the Davis reagent (2-(phenylsulfonyl)-3-phenyloxaziridine) or other similar oxazirid… | Wikipedia: Category:Name reactions | |
| Dess–Martin oxidation Oxidation | The Dess–Martin oxidation is an organic reaction for the oxidation of primary alcohols to aldehydes and secondary alcohols to ketones using Dess–Martin periodinane. | Wikipedia: Category:Name reactions | |
| DIALKYL MESOXALATES BY OZONOLYSIS OF DIALKYL BENZALMALONATES: DIMETHYL MESOXALATE (Propanedioic acid, oxo-, dimethyl ester) Oxidation | Preparation of Propanedioic acid, oxo-, dimethyl ester. Submitted by Lutz F. Tietze and Matthias Bratz1. DOI: 10.15227/orgsyn.071.0214. | Organic Syntheses | |
| Discussion Addendum for: (Oxidation of Nerol to Neral with Iodosobenzene and TEMPO) Oxidation | Preparation of Oxidation of Nerol to Neral with Iodosobenzene and TEMPO. Submitted by Francesca Leonelli1a, Roberto Margarita1b, and Giovanni Piancatelli*1c. DOI: 10.15227/orgsyn.089.0311. | Organic Syntheses | |
| Discussion Addendum for: Preparation of 4-Acetylamino-2, 2, 6, 6-tetramethylpiperidine-1-oxoammonium Tetrafluoroborate and the Oxidation of Geraniol to Geranial (2,6-Octadienal, 3,7-dimethyl-, (2e)-) Oxidation | Preparation of Discussion Addendum for: Preparation of 4-Acetylamino-2, 2, 6, 6-tetramethylpiperidine-1-oxoammonium Tetrafluoroborate and the Oxidation of Geraniol to Geranial (2,6-Octadienal, 3,7-dim… | Organic Syntheses | |
| Discussion Addendum for: (Synthesis of 1,2:4,5-Di-o-isopropylidene-D-erythro-2,3-hexodiulo-2,6-pyranose. A Highly Enantioselective Ketone Catalyst for Epoxidation/Asymmetric Epoxidation of trans-β-Methylstyrene and 1-Phenylcyclohexene using a D-Fructose-derived Ketone: (R,R)-trans-β-Methylstyrene Oxide and (R,R)-1-Phenylcyclohexene Oxide) Oxidation | Preparation of Synthesis of 1,2:4,5-Di-o-isopropylidene-D-erythro-2,3-hexodiulo-2,6-pyranose. A Highly Enantioselective Ketone Catalyst for Epoxidation/Asymmetric Epoxidation of trans-β-Methylstyrene … | Organic Syntheses | |
| Elbs persulfate oxidation Oxidation | The Elbs persulfate oxidation is the organic reaction of phenols with alkaline potassium persulfate to form para-diphenols. The reaction is generally performed in water at room temperatures or below, … | Wikipedia: Category:Name reactions | |
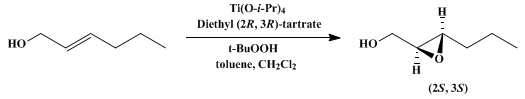
| ENANTIOSELECTIVE EPOXIDATION OF ALLYLIC ALCOHOLS: (2S,3S)-3-PROPYLOXIRANEMETHANOL (Oxiranemethanol, 3-Propyl-, (2S,3S)-) Oxidation | Preparation of Oxiranemethanol, 3-Propyl-, (2S,3S)-. Submitted by J. Gordon Hill1, K. Barry Sharpless1, Christopher M. Exon2, and Ronald Regenye2. DOI: 10.15227/orgsyn.063.0066. | Organic Syntheses | |
| ENANTIOSELECTIVE OXIDATION OF AN ALKYL ARYL SULFIDE: SYNTHESIS OF (S)-(-)-METHYL P-BROMOPHENYL SULFOXIDE Oxidation | Preparation of ENANTIOSELECTIVE OXIDATION OF AN ALKYL ARYL SULFIDE: SYNTHESIS OF (S)-(-)-METHYL P-BROMOPHENYL SULFOXIDE. Submitted by Carmelo Drago, Emma-Jane Walker, Lorenzo Caggiano, and Richard F. … | Organic Syntheses | |
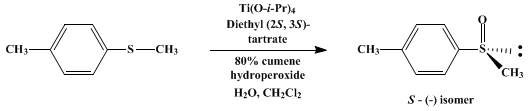
| ENANTIOSELECTIVE OXIDATION OF A SULFIDE: (S)-(−)-METHYL p-TOLYL SULFOXIDE (Benzene, 1-methyl-4-(methylsulfinyl)-, (S)-) Oxidation | Preparation of Benzene, 1-methyl-4-(methylsulfinyl)-, (S)-. Submitted by S. H. Zhao, O. Samuel, and H. B. Kagan1. DOI: 10.15227/orgsyn.068.0049. | Organic Syntheses | |
| EPOXIDATION OF OLEFINS BY HYDROGEN PEROXIDE-ACETONITRILE: cis-CYCLOOCTENE OXIDE (cis-9-Oxabicyclo [6.1.0] nonane) Oxidation | Preparation of cis-9-Oxabicyclo [6.1.0] nonane. Submitted by R. D. Bach and J. W. Knight1. DOI: 10.15227/orgsyn.060.0063. | Organic Syntheses | |
| Fétizon oxidation Oxidation | Fétizon oxidation is the oxidation of primary and secondary alcohols utilizing the compound silver(I) carbonate absorbed onto the surface of celite also known as Fétizon's reagent first employed by Ma… | Wikipedia: Category:Name reactions | |
| Fleming–Tamao oxidation Oxidation | The Fleming–Tamao oxidation, or Tamao–Kumada–Fleming oxidation, converts a carbon–silicon bond to a carbon–oxygen bond with a peroxy acid or hydrogen peroxide. Fleming–Tamao oxidation refers to two sl… | Wikipedia: Category:Name reactions | |
| Ganem oxidation Oxidation | In organic chemistry, the Ganem oxidation is a name reaction that allows for the preparation of carbonyls from primary or secondary alkyl halides with the use of trialkylamine N-oxides, such as N-meth… | Wikipedia: Category:Name reactions | |
| Griesbaum coozonolysis Oxidation | The Griesbaum coozonolysis is a name reaction in organic chemistry that allows for the preparation of tetrasubstituted ozonides (1,2,4-trioxolanes) by the reaction of O-methyl oximes with a carbonyl c… | Wikipedia: Category:Name reactions | |
| Hass–Bender oxidation Oxidation | In organic chemistry, the Hass–Bender oxidation (also called the Hass–Bender carbonyl synthesis) is an organic oxidation reaction that converts benzyl halides into benzaldehydes using the sodium salt … | Wikipedia: Category:Name reactions | |
| IN SITU CATALYTIC EPOXIDATION OF OLEFINS WITH TETRAHYDROTHIOPYRAN-4-ONE AND OXONE: trans-2-METHYL-2,3-DIPHENYLOXIRANE (Oxirane, 2-methyl-2.3-diphenyl-, trans-) Oxidation | Preparation of Oxirane, 2-methyl-2.3-diphenyl-, trans-. Submitted by Dan Yang, Yiu-Chung Yip, Guan-Sheng Jiao, and Man-Kin Wong1. DOI: 10.15227/orgsyn.078.0225. | Organic Syntheses | |
| Jacobsen epoxidation Oxidation | The Jacobsen epoxidation, sometimes also referred to as Jacobsen-Katsuki epoxidation is a chemical reaction which allows enantioselective epoxidation of unfunctionalized alkyl- and aryl- substituted a… | Wikipedia: Category:Name reactions | |
| Jones oxidation Oxidation | The Jones oxidation is an organic reaction for the oxidation of primary and secondary alcohols to carboxylic acids and ketones, respectively. It is named after its discoverer, Sir Ewart Jones. The rea… | Wikipedia: Category:Name reactions | |
| Juliá–Colonna epoxidation Oxidation | The Juliá–Colonna epoxidation is an asymmetric poly-leucine catalyzed nucleophilic epoxidation of electron deficient olefins in a triphasic system. The reaction was reported by Sebastian Juliá at the … | Wikipedia: Category:Name reactions | |
| Kornblum oxidation Oxidation | The Kornblum oxidation, named after Nathan Kornblum, is an organic oxidation reaction that converts alkyl halides and tosylates into carbonyl compounds. | Wikipedia: Category:Name reactions | |
| Lemieux–Johnson oxidation Oxidation | The Lemieux–Johnson or Malaprade–Lemieux–Johnson oxidation is a chemical reaction in which an olefin undergoes oxidative cleavage to form two aldehyde or ketone units. The reaction is named after its … | Wikipedia: Category:Name reactions | |
| Lindgren oxidation Oxidation | Lindgren oxidation is a selective method for oxidizing aldehydes to carboxylic acids. The reaction is named after Bengt O. Lindgren. | Wikipedia: Category:Name reactions | |
| METHYLTRIOXORHENIUM CATALYZED OXIDATION OF SECONDARY AMINES TO NITRONES: N-BENZYLIDENE-BENZYLAMINE N-OXIDE (Benzenemethanamine, (N-phenylmethylene)-, N-oxide) Oxidation | Preparation of Benzenemethanamine, (N-phenylmethylene)-, N-oxide. Submitted by Andrea Goti, 1 Francesca Cardona, and Gianluca Soldaini. DOI: 10.15227/orgsyn.081.0204. | Organic Syntheses | |
| MILD AND SELECTIVE OXIDATION OF SULFUR COMPOUNDS IN TRIFLUOROETHANOL: DIPHENYL DISULFIDE AND METHYL PHENYL SULFOXIDE ((Disulfide, diphenyl- and Benzene, (methylsulfinyl)-)) Oxidation | Preparation of (Disulfide, diphenyl- and Benzene, (methylsulfinyl)-). Submitted by Kabayadi S. Ravikumar, Venkitasamy Kesavan, Benoit Crousse, Danièle Bonnet-Delpon, and Jean-Pierre Bégué1. DOI: 10.15… | Organic Syntheses | |
| Oppenauer oxidation Oxidation | Oppenauer oxidation, named after Rupert Viktor Oppenauer, is a gentle method for selectively oxidizing secondary alcohols to ketones. | Wikipedia: Category:Name reactions |